4:57 PM Wed Feb 8 Tap here or pull up for additional resources At what temperature (in °C) would a 0.00510 M solution of glucose in water exhibit an osmotic pressure of 0.150 atm? O 14/ # F $ 4 D % 5 A Question 14 of 20 V ... & 7 * 8 9 1 1 4 7 +/- 0 D
4:57 PM Wed Feb 8 Tap here or pull up for additional resources At what temperature (in °C) would a 0.00510 M solution of glucose in water exhibit an osmotic pressure of 0.150 atm? O 14/ # F $ 4 D % 5 A Question 14 of 20 V ... & 7 * 8 9 1 1 4 7 +/- 0 D
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter6: Solutions And Colloids
Section: Chapter Questions
Problem 6.72P
Related questions
Question

Transcribed Image Text:4:57 PM Wed Feb 8
tab
At what temperature (in °C) would a
0.00510 M solution of glucose in water
exhibit an osmotic pressure of 0.150 atm?
Tap here or pull up for additional resources
caps lock
Q
A
W
S
#
3
E
D
$
4
R
%
5
F
T
^
6
Question 14 of 20
G
Y
&
7
H
U
8
J
I
9
K
1
0
1
4
7
+/-
0
L
P
Expert Solution
Step 1
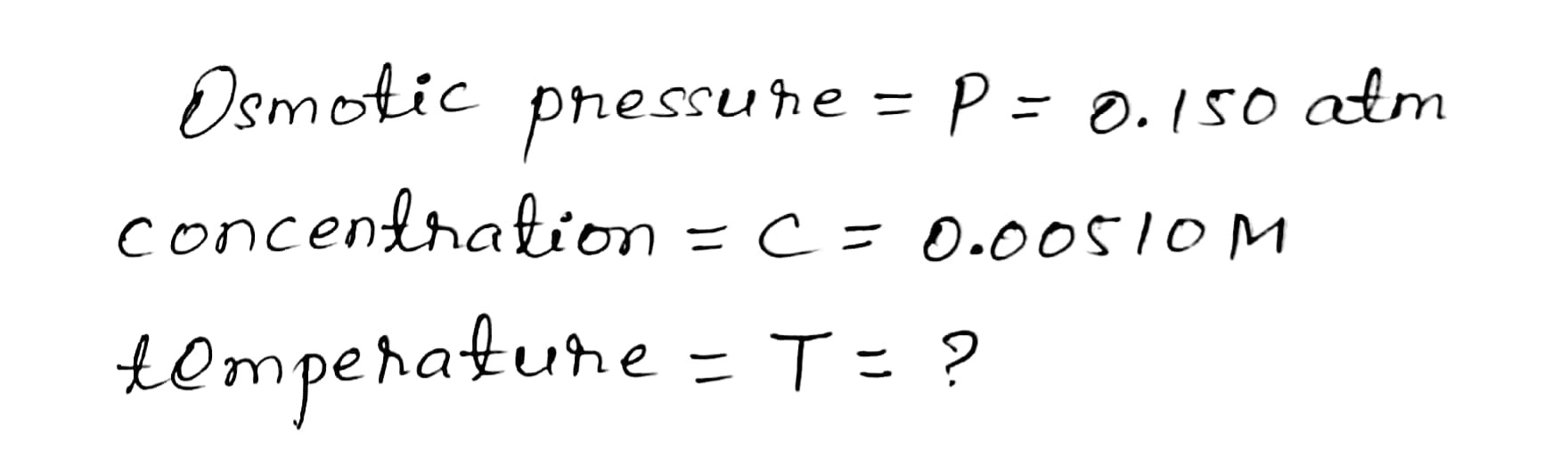
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning