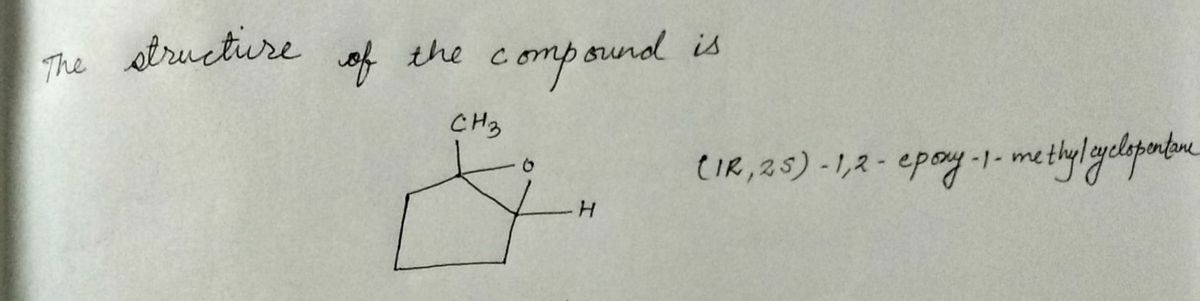
Draw the structural formulas for the first two intermediates formed in the hydrolysis of (1R,2S)-1,2-epoxy-1-methylcyclopentane with aqueous acid. Show stereochemistry if product is a meso compound.
Draw the structural formulas for the first two intermediates formed in the hydrolysis of (1R,2S)-1,2-epoxy-1-methylcyclopentane with aqueous acid. Show stereochemistry if product is a meso compound.
Chapter22: Carbonyl Alpha-substitution Reactions
Section22.SE: Something Extra
Problem 26MP: Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium...
Related questions
Question
100%
Draw the structural formulas for the first two intermediates formed in the hydrolysis of (1R,2S)-1,2-epoxy-1-methylcyclopentane with aqueous acid. Show stereochemistry if product is a meso compound.
Expert Solution
Step 1
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

