Q: Fresh pineapple will prevent the gelatin froms setting, but if I added canned pineapple into…
A:
Q: Describe how to isolate clove oil from the distillate?
A: To describe the method to separate the clove oil from the distillate.
Q: In the estimation of glucose with benedict's reagent, the solution must be continuously heated while…
A: Introduction : Benedict's test is a chemical test used to detect reducing sugars, this test uses…
Q: if you isolate more than one crop of crystal what must you do before combining them. (related to…
A: A widely adopted method in purifying compounds is recrystallization. In this method, the pure…
Q: If you use toluene as a solvent for recrystallization of biphenyl, what is the most likely reason it…
A: If use toluene as a solvent for recrystallization of biphenyl, it would be unsuccessful because…
Q: What is the benefit of using fluted filter paper instead of not folding it?
A: “Fluted “ filter paper (LEFT) is used when we wish to separate a liquid and a solid, keeping the…
Q: A mixture is made up of low molecular weight carboxylic acid, alcohol and ester. What one step…
A: Extraction is the method to separate components from mixture
Q: Why is methanol chosen as the solvent for recrystallization? A. Because it can easily be boiled…
A: Solvent used in recrystallization should have below properties: At high temperatures, it should be…
Q: 18. For an extraction to be successful, the organic layer should be immiscible or miscible with the…
A: In extraction process, organic and aqueous solutions are thoroughly mixed with solute and is allowed…
Q: What is the purpose of the filter paper moistened with ammoniacal silver nitrate solution in the…
A: The functional groups are the key components associated with a molecule that is highly responsible…
Q: The Coca- cola is diluted and reacted with molybdate/metavanadate reagent before the %T is measured.…
A: Phosphoric acid (H3PO4) is used primarily in the manufacture of fertilizers, detergents and…
Q: 18g ethanoic anhydride
A: Molecular weight for ethanoic anhydride=102 gm Given weight=18 gm
Q: Acetone will give no reaction with sodium bisulfite. Yellow crystals are formed in the iodoform test…
A: Carbon centre in acetone is electrophilic and it can give nucleophilic addition reaction.
Q: A fellow student somehow mixed an aspirin sample with paracetamol after synthesising both. Explain…
A: The functional groups are the key components associated with a molecule and they can furnish the…
Q: Which of the ff is not true about recrystallization? a. acetanalide recrystallized only at cold…
A: Recrystallization is done to purify the compounds so as to get the purest form.
Q: Measure the weight of acetylsalicylic in aspirin. note: % of acetylsalicylic acid in aspirin is 5%…
A:
Q: When choosing a recrystallization solvent, what properties must be considered? a. Its boiling…
A: The solvent should boil in the range 50–120°C. Impurities should either be insoluble in the hot…
Q: If the aspirin crystals were not completely dried before the melting point was determined, what…
A: Freezing point depression or melting point depression occurs when a solute (or impurity) is added to…
Q: theoretical yield for reaction theoretical mass for stilbene, acetic acid, and pyridinium tribomide…
A: From the reaction it is clear that one mole of stilbene reacts with one mole of Br2 in presence of…
Q: Explain why diester A is now often used as a plasticizer in place of dibutyl phthalate
A:
Q: 1 Explain the comparative reactivity and hazards of NABH. as compared to LIAIH. Also, compare the…
A: Lithium aluminium hydride and sodium borohydride are popular reducing agents.
Q: Compound X is quite soluble in toluene, but only slightly soluble in petroleum ether. How could…
A: Given, Compound X is quite soluble in toluene, but only slightly soluble in petroleum ether.
Q: Analyze the IR spectrum of acetic acid.
A:
Q: A. Type of Test B. POSITIVE RESULT C. SUBSTANCES THAT 1. lodoform Test A. white precipitate WILL…
A: Qualitative analysis of compounds:
Q: ADDED REAGENT(S) OBSERVATIONS Ba(NO3)2 NVR HNO3, Fe(NO3)3 Blood red aqueous layer; pink toluene…
A: Anions can be detected by various reagents such as ferric nitrate, potassium permanganate and barium…
Q: Why did sucrose give a positive test with Benedict’s solution after heating?
A: Benedict's reagent is a complex mixture of sodium carbonate, sodium citrate and copper(II) sulfate…
Q: Explain how the effectivity of 70% alcohol as disinfectant compared to the action of 40% and 95%…
A: The mode of action as antimicrobial agents depends on its concentration. Alcohol concentrations of…
Q: Why is it important to collect the distillate into a test-tube that is immersed in an ice-bath? To…
A:
Q: What are some reasons why the melting point might be lower than the literature value? (Hint: Assume…
A: A pure compound generally melts over a range of temperature. If the melting point of the sample is…
Q: Cinnamaldehyde is a naturally occurring compound present in cinnamon. Cinnamaldehyde can be…
A:
Q: What happens when benzaldehyde reacts with sodium bisulfite? What happens when acetone reacts with…
A:
Q: Measure the benzene and water in the unknown solution mixture using Ultraviolet–visible spectroscopy…
A: Uv-vis spectroscopy
Q: which has a positive result in ninhydrin test? a. ethyl ethanoate b. sucrose c. proline d. 1-butanol…
A: Amines - These are the compounds containing the NH2 group. Amino acids - These are the organic…
Q: 3.5 mmol of 4-(Dimethylamino)benzaldehyde + 3.5 mmol of 4′-Methylacetophenone + 2 mL of 95% ethanol.…
A: Limiting reagent: The reagent which is completely consumed during the reaction. Or we can say the…
Q: caffeine
A: The crude caffeine contains contaminates, usually other organo compounds that were part of the plant…
Q: What forms from the substance of mixed with chloroform with a presence of KOH
A:
Q: What is the purpose of Chromic acid test? a. What are the reagents used? b. Write oxidation…
A: The Jones reactant is used to oxidise aldehydes and alcohols while reducing chromic acid, producing…
Q: Write the observations and results for the following reactions: A. Hinsberg Test of Aniline B.…
A:
Q: Why is the percentage of acetic acid in date vinegar more than apple cider vinegar?
A: Both vinegars are produced by fermentation. in the formation of date vinegar, the sugar is converted…
Q: 120. This method is used to isolate phenol from biological material: A) Mineralization B) Extraction…
A: Isolate of phenol from biological material is a separation process. In the separation process…
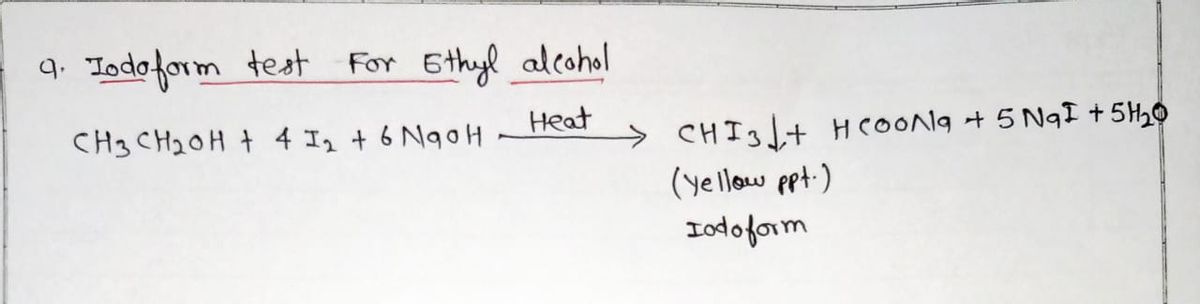
Q: what odor does ethyl alcohol produces in iodoform test?
A: What odor does ethyl alcohol produces in the iodoform test?
Q: How many grams of the excess reagent left unreacted?
A: Given :- Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g) Amount of Mg = 2.50 g Molarity of HCl solution =…
Q: Answer True or False Benzoin give positive test with Fehling's reagent. Solubility of aniline in…
A: Answers in step 2.
Q: Given a sample of anise oil for its phenol (anethol) content determination, 10 ml was used as sample…
A: Percent composition of an compound is defined as the amount of the that compound divided by the…
Q: Explain the Sodium Bisulfite Test for aldehydes (positive and negative result)
A: When Aldehydes react with sodium bisulfite (NaHSO3), then a water soluble crystalline solid…
Q: Caused by the severe dehydration effect of formaldehyde on tissue A. fumigation B.…
A: Caused by the severe dehydration effect of formaldehyde on tissue A. fumigation B.…
Indicate the expected result when the iodoform test is done on:
a. Ethyl alcohol
b. Isopropyl aclcohol
c. t-Butyl alcohol
d. Phenol
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- CLASSIFICATION OF ORGANIC COMPUNDS. USING SOLUBILITY EXAMPLE ( ANILINE + WATER = INSOLUBLE) SOLVENT LITMUS PAPER TEST sample 5% NaHCO3 5%HCL 5%NAOH CONC. H2SO4 ETHER RED LITMUS PAPER RESULT BLUE LITMUS PAPER RESULT CLASSIFICATION ANILINE ACETIC ACID PHENOL CYCLOHEXANOL 2-PROPANOLOL BROMOBENZENE ETHYLENE GLYCOL BENZOIC ACIDMULTIPLE CHOICE 1. A 37-40% solution of formaldehyde. * A. butyraldehyde B. acetone C. formalin D. acetaldehyde 2. Which of the following will GIVE a positive reaction to haloform reaction? * A. propanal B. butanal C. pentanal D. acetaldehyde1. Cite some reactions in which formaldehyde behaves differently from other aldehydes. 2. Give some biological and medical applications of: a. formalin b. chloral c. urotropine
- MULTIPLE CHOICE 1. What is the color of the solid produced when acetaldehyde is added with saturated Sodium bisulfite? A. yellow solid B. silver mirror C. brick red precipitate D. white crystalline precipitate 2. An aldehyde that is used in silvering mirrors. * A. propionaldehyde B. formaldehyde C. acetaldehyde D. butyraldehydeWhat kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care products and their function is to soften skin cells and to lessen wrinkles? A. Ethly acetate B. Alpha hydroxyl acids C. Phenols and phenol derivatives D. Aliphatic alcoholswhy ethyl acetate is immiscible with water?
- MCQ 35: Halogens dissolve well in A. pentane B. ethane C. cyclohexane D. waterGrignard reagents react with aldehyde to form: а. Carbon dioxide b. Secondary alcohol С. Ketone d. Carboxylic acid.. .is the metal precipitated in Tollen's or Mirror's test for benzaldehyde. (e a. Na-metal O b. Ag-metal c. Hg-metal O d. Al-metal e. Cu metal
- Multiple matching type. Match the following organic compounds with the correct classification of in the second column and with it's use in third column. Write the letter of the correct answer in the space allotted. Separate the two letters with a hyphen, example: A-M. ANSWER ORGANIC COMPOUND CLASSI FICATION USE Barbiturates K. Thiol A. Local anesthetic Paracetamol L. Thioether B. Immunosuppressant Nicotine M. Amine C. Depilatory agent Azathioprine N. Amide D. Stimulant Cimetidine E. Antipyretic Gluthathione F. Allergic response Histamine G. Sedative Lidocaine H. Anti ulcerative Clindamycin I. Antioxidant Epinephrine J. Antibioticwhich has the greatest acidity? a. 2-hexanol b. valeric acid c. naphtol d. ethyl propionate which produces effervescence when dissolved in 10% sodium bicarbonate? a. 2-hexanol b. valeric acid c. napththol d. ethyl propionateGive the products formed when benzaldehyde and benzoic acid are treated with the given reagents. a. Tollen’s reagentb. phenylhydrazine, H+c. HCNd. NH2OHe. 1 mole H2, Nif. 1 mole CH3OH, H+g. LiAlH4 then H2O, H+h. 2 moles CH3OH, H+i. CH3MgCl, then H2O, H+j. H2O

