Make a model of each compound, draw it in its most symmetric conformation, and determine whether it is capable of showing optical activity. cis-1,3-dibromocyclohexane
Make a model of each compound, draw it in its most symmetric conformation, and determine whether it is capable of showing optical activity. cis-1,3-dibromocyclohexane
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter2: Alkanes And Cycloalkanes
Section: Chapter Questions
Problem 2.35P: Consider 1-bromo-2-methylpropane and draw the following. (a) The staggered conformation(s) of lowest...
Related questions
Question
Make a model of each compound, draw it in its most symmetric conformation, and determine whether it is capable of showing optical activity.
cis-1,3-dibromocyclohexane
Expert Solution
Step 1
Asymmetric centre –
- Asymmetric centre is a molecule having a special arrangement of atoms which is not superiposable on its mirror images.
- So, molecule consists of asymmetric (Chiral) carbon atom called as asymmetric centre.
- Chiral carbon atom is a Carbon which bears four different groups (non identical substituents).
- By using these criteria chiral carbon can be predicted from given molecules.
- Molecule with chiral carbon that is asymmetric centre is optically active.
Plane of symmetry:
The plane in molecule which split molecule in to two equal halves.
This is an imaginary plane.
Step 2
Answer of given question:
Structure of cis-1, 3-dibromocyclohexane is as shown below:
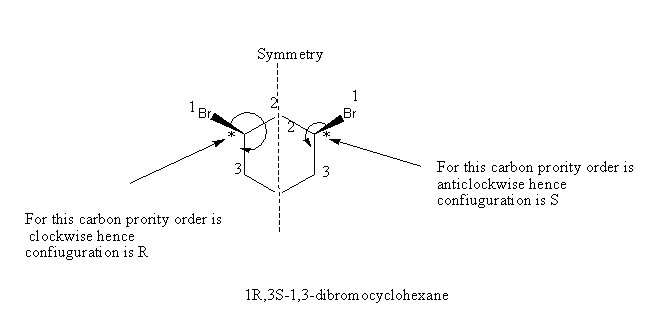
Beyond that this compound has two chiral carbons but it will not show the optical activity as it has a plane of symmetry in the molecule. Hence cis-1,3-dibromocyclohexane is optically inactive.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
