Q: GHN, O
A: Given equation is : C11H8N2O + C7H7N3O3 -----------> C18H13N5O3 Balance the equation = ?
Q: 1. How many MOLECULES of boron tribromide are present in 2.51 moles of this compound ? molecules. 2....
A: Number of moles is calculated by dividing given mass with molecular mass.
Q: An analyst weighs the same tablet three times on a well calibrated analytical balance and obtains th...
A: Environmental changes, such as in temperature or humidity. Exposure to harsh conditions, such as cor...
Q: pH =
A:
Q: NH3, Is made industrially by the Haber process. This is an equilibriur reaction. N2(g) + 3H2(g) =2NH...
A:
Q: 7.11x +.15 =.975-2 7 Calculate the equilibrium concentrations of all species if 3.000 moles of H2 an...
A:
Q: Classify each of the following compounds as a strong acid, weak acid, strong base, or weak base, and...
A: The strength of the acids or bases can be explained with the help of dissociation constant of an aci...
Q: 1. A galvanic cell is set up by immersing a silver electrode in 1.0 M silver ion solution to form on...
A: Anode is the electrode on which oxidation takes place and Cathode is the electrode where reduction t...
Q: What is the net ionic equation for the following reaction? What two ions form the precipitate here. ...
A: An aqueous solution of sodium carbonate reacts with an aqueous solution of iron(II) chloride to form...
Q: Give the five examples for glass forming materials. What is the role of modifiers in glass making. w...
A: a) Glass Forming Material - The Glass Former is the element that actually makes the glass (glaze) on...
Q: 5. Identify the index of plane for the given unit cells below. 0,0,0 0,0,0 (a) (b) 1/2 1/4
A:
Q: Consider the following reaction: UO22* a) + Pbis) + 4H*taq) U4+ (aq) + Pb2*(aqg) + 2H2Ow Which speci...
A: Given , Reaction : UO22+(aq) + Pb(s) +4H+(aq) →U4+(aq) +Pb2+(aq) +2H2O(I)
Q: Compute the 6 mass of a solution containing 5g sodium chloride in 150g solution? What is the molalit...
A: Molality: no of moles of solute present in one kg of solvent is called molality of the solution. Mol...
Q: What mass of magnesium contains the same number of particles as there are in 6g of carbon 12
A: Given, Mass of C = 6g What mass of magnesium contains the same number of particles as there are in...
Q: A freshly isolated sample of 90Y was found to have an activity of 2.5 ×105 disintegrations per minut...
A: Given, A freshly isolated sample of 90Y was found to have an activity of 2.5 ×105 disintegrations pe...
Q: a) Determine which of the two reactants (MgCO34) or HCla) is the limiting reagent. b) Determine how ...
A: Reaction MgCO3 (s) + 2HCl(aq) ------> MgCl2 (aq) + H2O (l) + CO2 (g); ∆Hrxn0...
Q: mass
A:
Q: Explain metal degredation and ceramic degredation briefly with the help simple drawings?
A: Given below
Q: O Represent the following equations with an appropriate diagram/illustration/ or model. Indicate a l...
A: Answer is below
Q: (van’t hoff) use van’t hoff to calculate freezing and boiling points; don’t know what went wrong
A: Vant Hoff factor,i = 1.83 Molality of solution = 7.75 × 10-2 M Kf = 1.86 °Ckg/mol Kb = 0.512 °Ckg/m...
Q: a. Mole Fraction of H2O2 b. Molarity
A:
Q: Define electrolysis. A)Process using chemical energy to cause a nonspontaneous process to occur B)Pr...
A:
Q: O2 (g) CO: (g) H:O (1) CH6 (1) ->
A:
Q: What product is formed when the alcohol is oxidized with K2Cr2O7? In some cases, no reaction occurs ...
A:
Q: NChane gar oscuples a wume of12iters at220 Boyle's Law. thkrine oar rcudes n wme of 12hters at 720 O...
A: According to Boyle's law ' volume occupied by a gas is inversely proportional to the pressure of gas...
Q: Question 2 The concentration of a diluted "wine" sample was found to be 0.32 %(v/v) ethanol. Assumin...
A: Answe is attached below
Q: Which of the following are typical impurities in active pharmaceutical ingredients? I. intermediate...
A: An impurity in pharmaceuticals is classified as any component that is not the entity defined as the ...
Q: Question 10 Consider placing a piece of iron, Fes, into a 1 M solution of cadmium ion, Cd2* ag). Sel...
A:
Q: Consider the balanced equation of KI (molar mass=166.0 g/mol) reacting with Pb(NO3)2 to form a preci...
A: In the given question, we have the following balanced reaction equation: 2KI(aq)+Pb(NO3)2(aq)→PbI2(s...
Q: Just nitrosyl bromide NOB1 (0.64 mol) was placed in a 1.00 L flask. When heated it readily decompose...
A: Nitrosyl bromide, NOBr decomposes into nitrogen monoxide, NO, and molecular bromine, Br2. The equili...
Q: The reaction between iron(II) (Fe2+) and dichromate (Cr, 0-) in the presence of a strong acid (H*) i...
A:
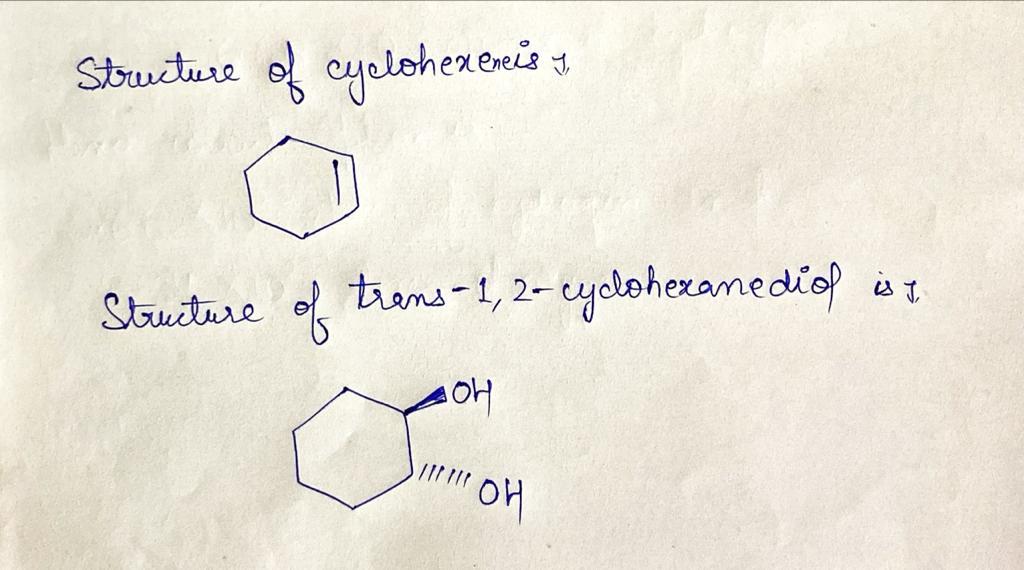
Q: 1) Chapter 2 warm-up: What is the lowest energy conformation of each molecule below? Justify your an...
A:
Q: What is oxidiation number?
A: Oxidation number: it is defined as the charge (real or imaginary) when an atom appears to have when ...
Q: What is the boiling point of a solution made by dissolving 10.0 g sucrose ( C12H22O11C12H22O11 ) in ...
A:
Q: Which of the following are typically included in a Pharmacopoeial monograph? I. Tests to confirm the...
A:
Q: Identify (circle) the strong acids from the list below: H,SO, HNO, HCIO, HCI HCIO, H,S Identify (cir...
A: Acids are those substance which releases H+ ion when dissolved in water while base are those substan...
Q: .Calculate the degree of reduction for NH3 and N2 on both an NH3 basis and an N2 basis (write units)...
A: In a chemical reaction that involves electron transfer, one of the species gains electrons while the...
Q: If the pH of a solution is 0, which of the following is true? [H+] = 10 -14 M, [OH-] = 1 M ...
A: Which of the following is correct...
Q: Question 1 For the reaction below, which species is being oxidized? H2 + Cl2→ 2 HCl H2 Cl2 HCl Quest...
A: This question belong to Redox Reaction. Oxidation : Oxidation means increase in oxidation state in t...
Q: Cyclopropane, C3H6, is converted to its isomer propylene, CH2=CHCH3, when heated. The rate law is fi...
A:
Q: 5.20 Name each of the following compounds according to substitutive IUPAC nomenclatu (а) (CH3)2СНCН,...
A: We have to correct the IUPAC NAME of the following compound. Some points necessary while writing IUP...
Q: In the following reaction, potassium chlorate decomposes to potassium chloride and oxygen gas. How m...
A:
Q: Which is the more effective way to increase the thermal efficiency of a Carnot engine; to increase T...
A: A Carnot cycle taking place between a hot reservoir at temperature Th and a cold reservoir at temper...
Q: You calculate an interference value of 0.59. What does this number tell you? a. You will not observ...
A: Crossing over does not occur uniformarly.formation of one chiasma reduced the chances of formation o...
Q: A gas occupies 3.5 L at standard pressure. Find the volume of the gas when the pressure is 1140 mm ...
A:
Q: • Predict the major product of the following reactions: H,C. CH H,C CH H,C H,C H,C. , AICI, H,C CH H...
A: fridel craft reaction: Friedel-craft alkylation refers to the replacement of an aromatic proton with...
Q: Calculate the pOH and the pH of a 0.400 M Ba(OH), solution.
A: We have to predict the pOH and pH of solution.
Q: Assuming that the analyte in A and B are the same organic molecule, select all possible correct reas...
A: The correct option is concentration differences.
Q: reduced?
A:
Q: Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid...
A:

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Predict the major products. C,H5-C-CH; а. 1/2 b. (CH;),CH-C-Ħ#", heat (CH),СH-C-Нт C,H5-CH2-C-Ħ с. d. C,Hs-C-CH; H" CaH-C-CH С,Н-С-СH е. f. CHs-C-CH;¬N#OH CH;-C= C-CHOTE gSO g. h. C,Hs-C-C,H5 H*6. The product(s) of the following reaction dd blow di CH2-CH2 excess HBr is/are: CH2 CH2 heat Od Al () (A od Ocf D (CHPPCOK (CHPCON G)D CH;CH2OCH,CH3 HO CH;CH,CH,CH,OH and CH;CH,CH2CH,Br II H t mol boumol toubong soinm od o CH2-CHBR HO CH2 CH2 BrCH-CH,CH-Cн,ОН and BrCH,CH,CH-CH,Br CityCHCH COM III IV A) I B) П O II D) IV E) None of these HO CH CH'OHWhich of the following provides a synthetic route to convert 3-iodo-2-methyl-1- butene into 2-methyl-2-butene? 1. NaOH; 2. H₂, Pt 1. H₂, Pt; 2. Br2 1. H₂, Pt; 2. NaOEt 1. H₂, Ni₂B; 2. KOtBu 1. H₂, Pt; 2. KOtBu
- 1. Identify the products of the following transformations. Br HCI CH; H;C-c=CH, 1. BH3 2. HO, H;0,, H,0 H*, H;O Br2 H,O(a) 2-methyl-2-pentanol Draw the structure of the alkene that was used to prepare the alcohol in highest yield. AAVIL ? ChemDoodleⓇ Which process does this employ? 1. Hg(OAc)2, H₂O; 2. NaBH4 01. BH3; 2. H₂O2, NaOH OsO4, H₂O2 On n [ ]#23. The conversion of cyclohexene to cyclohexane requires which reagent and/or conditions given as responses? Оа. НC O b. H20 and heat and pressure Oc. H20 and H2SO4 O d. H2 and Pt O e. no correct response is given
- to this answer, estion 54 A cycloalkene having only one cartion.carbon double bond will have the general formula CnH2n-4 CnH2n 2 CnH2n CnH2h+2 AMoving to the dext questioni prevents chaniges to this answer. chapter10eted ppt Open tile chapter18 egitnd(1).ppt Ciosn file |||5. Cychonexene dnd KMNQ4 In 6.Onknown 2 Calkene) reactants. and KMn to OH Mr Products 7. Gyclonexene and Hz SQu H-O to 1.Rank the compounds by DECREASING reactivity towards nitration with HNO3/ H2SO4 (most reactive on the left). NO2 CH3 II III IV O A. V>V>I> || > || O B. V>I> III > || > IV oCV> || >1> V> I| O D. I > V> > || > IV O E. II > V >1> [IV >| Moving to another question will save this response. Co search
- 39. Which of the following reactions will produce the highest percentage yield of 1-butene. C,H5OK*/C,H;OH A. CH;CH2CH(N*(CH3);)CH3 alc. KOH B. CH3CH2CHCICH3 conc. H2SO4 С. CH;СНОНСH-CH; heat NaOC,H5 D. CH3CHBrCH2CH3 СН,ОНapp.101edu.co oudy @ 2 X V * UL Week 7: Panopto ||| 3 D E PII $ 4 R Rank these alkyl halides in order of increasing reactivity in an SN2 reaction. F 4x X UL LTU 7-1A F4 HHH % 5 T G 4, CI F5 A 6 4) Y X Question 16 of 24 F6 C|Chegg.com & 7 U F7 ★ A) III < | < || 8 B) || < | < ||| C) ||| < || < | D) || < ||| < | E) | < ||| < || PrtScn 1 FB X + Home ( 9 F9 O ) End 0 D 67 ENG F10 P PgUp Tp S ( D [ * O 6:15 PM 10/18/2022 PgDn + F12 0 SL 3What is the organic product formed in the following reaction? NaOC₂H₂ H+(aq) стеносли + OC₂H HOC₂H | -CH₂CH = ||| Select one: A. II B. I C. IV D. III ° -CH₂CH₂ OC₂HS IV -CH OC₂H₂ CH3 OH CH3 O OC₂H₂ bc₂Hs

