Thermodynamic Properties of HFC-134a Refrigerant (1,1,1,2-tetrafluoroethane) CH2FCF3 molecular Weight 102.3 Bolling point at one atmosphere -26.06C Critical Temperature 374.23K Critical Pressure 4060.3 kPa Critical Density 515.3 kg/m^3 Critical Volume 0.00194 m^3/kg 1) The condenser of a home refrigerator is commonly underneath the appliance, thus the condensing refrigerant exchanges heat with household air, which averages 70oF. It is proposed to reconfigure a refrigerator so that the condenser is in the garage, where the average yearly temperature is about 50oF. Assume a freezer temperature of 0oF, a minimum temperature difference of 10K for heat transfer, and an actual coefficient of performance of 60% that of a Carnot cycle. Calculate the Coefficient of Performance for both configurations and discuss the pros and cons of this proposal. 2) Which is the most effective way to increase the coefficient of performance of a Carnot refrigerator: to increase TC with TH held constant or to decrease TH with TC held constant? Justify mathematically. For a real refrigerator, do either of these strategies make sense?
CH2FCF3
molecular Weight 102.3
Bolling point at one atmosphere -26.06C
Critical Temperature 374.23K
Critical Pressure 4060.3 kPa
Critical Density 515.3 kg/m^3
Critical Volume 0.00194 m^3/kg
1) The condenser of a home refrigerator is commonly underneath the appliance, thus the
condensing refrigerant exchanges heat with household air, which averages 70oF. It is proposed to
reconfigure a refrigerator so that the condenser is in the garage, where the average yearly
temperature is about 50oF. Assume a freezer temperature of 0oF, a minimum temperature
difference of 10K for heat transfer, and an actual coefficient of performance of 60% that of a
Carnot cycle. Calculate the Coefficient of Performance for both configurations and discuss the
pros and cons of this proposal.
2) Which is the most effective way to increase the coefficient of performance of a Carnot
refrigerator: to increase TC with TH held constant or to decrease TH with TC held constant? Justify
mathematically. For a real refrigerator, do either of these strategies make sense?
Case1:
when the refrigerator is placed in household air,
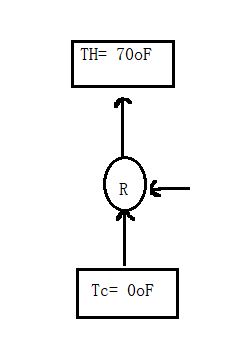
TH= 70F= 294.26 K
TC = 0F= 255.372 K
Carnot COPC= TC/(TH-TC) = 6.566
Case2:
when the refrigerator is placed in garage,
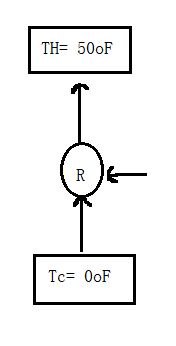
TH= 50F= 283.15 K
TC = 0F= 255.372 K
Carnot COPC= TC/(TH-TC) = 9.193
COP in case 2 is greater than COP in case 1,
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images









