
Concept explainers
(a)
Interpretation: Structure of the major product expected from the ionic addition of
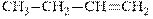
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
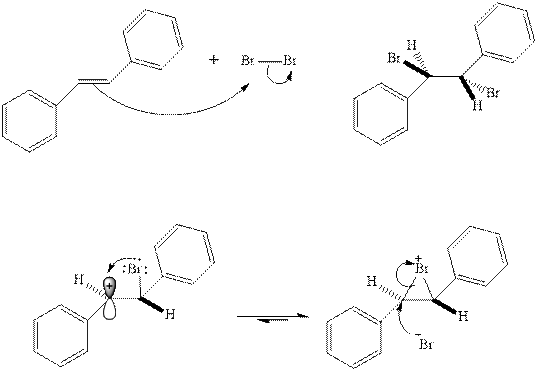
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
(b)
Interpretation: Structure of the major product expected from the ionic addition of
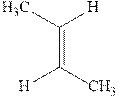
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
(c)
Interpretation: Structure of the major product expected from the ionic addition of
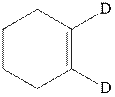
Concept introduction:Alkenes are considered electron-rich and undergo bromination addition reaction with
The mechanism of
In the second step, the strained bromonium ion intermediate opens while
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- 5B In the following reactions, mixtures of alkenes and ethyl ethers are formed. Draw their structures. Explain which is or are likely to be the main product(s) in each reaction. In case of formation of two isomers of alkenes, explain which is formed in greater proportion CH3 CH3 H3C-C H -Br CH3 EtOHarrow_forwardWhat are the products formed from the following reactions: 1) The addition of dimethylborane (Me2BH) to ethylene 2) The addition of dimethylborane (Me2BH) to acetylene Please include the formulas for the products as well. Thank you!arrow_forward∆H° values obtained for a series of similar reactions are one set of experimental data used to determine the relative stability of alkenes. Explain how the following data suggest that cis-but-2-ene is more stable than but-1-ene (Section 12.3A).arrow_forward
- Which alcohols can be prepared as a single product by hydroboration– oxidation of an alkene? Which alcohols can be prepared as a single product by the acid-catalyzed addition of H2O to an alkene?arrow_forwarddraw the two possible carbocations that can form when this alkene reacts with a strong acid (such as HBr or H3O+). of the two structures you drew, circle the more stable carbocationarrow_forwardA hydrocarbon of unknown structure with the formula CgH14 was isolated. In an effort to determine the structure, the compound was subjected to hydrogenation with H2 and Pd, which afforded a product with a formula of C8H16 Which of the following compounds are possible candidates for the starting hydrocarbon?@GMU 2020 Me II II Mearrow_forward
- a) What products would you expect from the elimination reaction of 3-Bromo-2- methylpentane? Show the reaction by writing the condensed structural formula of the reactants and products. Identify the major and minor products. b) What alkyl halide might the 3,6-Dimethyl-1- heptene have been made from?arrow_forward5 Write the molecular formula for each alkane. Y \ y t (a) (c) HE8 iw 146 (b)- eriam in Beris mal bond HO H ox dT O HOH w ar HO HO (s) HOarrow_forwardWhat is the molecular formula of the following alkene? CI o CH14CI o CH13CI o CH15CI o CH11CIarrow_forward
- Which products are formed when hydrobromic acid is added to (a) trans-2-hexene, (b) 2-methyl- 2-pentene, and (c) 4-methylcyclohexene, and how many regioisomers can be formed in each case?arrow_forwardDraw the structure of the reactant and name the product of the following reactions: 3-methyl-2-heptanone + NaBH4arrow_forwardΔH° values obtained for a series of similar reactions are one set ofexperimental data used to determine the relative stability of alkenes. Explain how the cis-but-2-ene is more stable than but-1-enearrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning


