
Concept explainers
Interpretation: To propose a plausible synthesis for
Concept introduction: The retrosynthesis begins with disconnection at the ether groups, at two of the
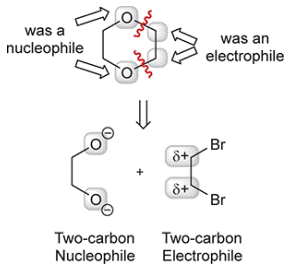
Each of these starting materials can be made from acetylene.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
- 1. Predict the product, and propose a mechanism for the following nucleophilic addition to aldehyde or ketone. Are the products different from one another? Why do the reactions proceed via different mechanisms? Note the charges on the intermediates of acid and base catalyzed reactions. Oxygen nucleophile in acidic condition H2O, [H+] Oxygen nucleophile in basic condition NaOHarrow_forwardIF esize this compound by the → t-butyl ethyl ether Show the steps necessary to synthesize this compound by a rignard reaction. Start with an alkyl halide; after that you can add any organic or inorganic compound. → 1-hexanol Consider the following compounds:arrow_forwardWrite an equation for the acid-base reaction between 2,4-pentanedione and sodium eth- oxide and calculate its equilibrium constant, K. The pK, of 2,4-pentanedione is 9; that of ethanol is 15.9. CH,CCHÖCH, + CH,CH,O Na* H 2,4-Pentanedione Sodium ethoxidearrow_forward
- O NaBH4 Synthesis #2: Select a different set of two substrates and one reagent that could be combined to prepare benzphetamine. tymen Ph Ph Ph Ph NH Ph H O NaBH3CN, H+ 20= ano [H*], NaBH-CN Nucleophilearrow_forward2 By using the same alkene, how do you synthesize the following alcohols? Give the alkene and show the mechanisms for each product formation. OH OH НО.arrow_forwardPropose an efficient synthesis for two of the following target molecules, starting with any of the following starting materials for the source of carbon atoms: (i) benzene, (i) any alcohol containing four or fewer carbon atoms, (im) carbon dioxide, (iv) cyanide ion. This means that any carbon atom that ends up in the target molecule must come from one of these starting materials! Br (а) H2N (b) HOarrow_forward
- CH3 གོན་ནི། ཡོན་ Br₂, H₂C CH₂Br H3C CH3 H₂C CH3 Aldehydes and ketones can be halogenated at their a-position by reaction with Cl₂, Br2, or 12, under acidic conditions. Using Br₂ under acidic conditions, an intermediate enol is formed which adds bromine at the a-position. The reaction stops after the addition of one bromine because the electron-withdrawing halogen decreases the basicity of the carbonyl oxygen, making the protonation less favorable. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions :0: :OH н-он H-OH₂ H₂O H₂C. H3C. CH3 CH3 H3C CH3 H3C CH3 Σαarrow_forwardPropose a structure for an aromatic hydrocarbon, C10H14, that can form only one C10H13Cl product on substitution of a hydrogen on the aromatic ring with chlorine.arrow_forwardWrite the Ky reactions for pyridine, for sodium 2-mercaptoethanol, and for cyanide. H+ binds to S in sodium 2-mercaptoethanol and to C in cyanide. (Hint: Spectator ions, which do not react in solution, appear on both sides of the reaction and can be removed.) N: HOCH.CHS: Na+ CN Pyridine Sodium 2-mercaptoethanol Cyanide Refer to the Ka values below for their conjugate acids. Which of the three bases shown above is the strongest? NH HOCH.CH.SH HCN Pyridinium ion K. = 6.3 x 10-6 2-Mercaptoethanol K. = 1.9 X 10-10 Hydrogen cyanide K = 6.2 x 10-10arrow_forward
- Alcohols can act as either acids or bases, similar to water. Draw the products of the following reaction: H3C-OH + Na+NH2- You should include all products. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher.arrow_forwardi) ii) 1. Propose a mechanism for the following reactions. H (CH3)2NH H* NaOH EtOHarrow_forwardStarting from acetylene and alkyl halides not longer than 4 carbon atoms, show the synthesis of 3-octanol and a ketone. Indicate the reagents in each step.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

