
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14.23, Problem 26P
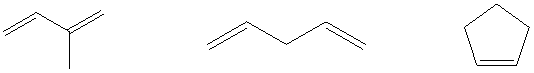
Which one of the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What wavelength is the peak absorbance of CoCl2?
what is the constitutional isomer for this spectrum? (C5h12O)
(b) Determine the topicity of the groups or
atoms indicated by arrows :
Ph
H<
(i)
H<
H3CO
OCH3
H
focecomt.
A
(ii)
CH3
CH3
Chapter 14 Solutions
Organic Chemistry - Standalone book
Ch. 14.3 - Prob. 1PCh. 14.3 - Prob. 2PCh. 14.4 - The 1H NMR signal for bromoform (CHBr3) appears at...Ch. 14.5 - identify the most shielded and least shielded...Ch. 14.5 - (a) Assign the chemical shifts 1.6, 2.2, and 4.8...Ch. 14.5 - Assign the chemical shifts 1.1, 1.7, 2.0, and 2.3...Ch. 14.5 - Assign the chemical shifts 1.6, 4.0, 7.5, 8.2, and...Ch. 14.6 - The 300-MHz 1H NMR spectrum of 1,4-dimethylbenzene...Ch. 14.6 - Prob. 9PCh. 14.6 - How many signals would you expect to find in the...
Ch. 14.7 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.8 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.11 - Prob. 13PCh. 14.11 - Prob. 14PCh. 14.12 - Hydrogen bonding between the oxygen of dimethyl...Ch. 14.14 - Prob. 16PCh. 14.15 - The 13C NMR spectrum of 1-bromo-3-chloropropane...Ch. 14.15 - Consider carbons x, y, and z in p-methylanisole....Ch. 14.15 - Prob. 19PCh. 14.16 - To which of the compounds of Problem 14.16 does...Ch. 14.18 - DEPT spectra for a compound with the formula...Ch. 14.20 - Vibrational frequencies are sensitive to isotopic...Ch. 14.21 - Prob. 23PCh. 14.22 - Prob. 24PCh. 14.23 - Prob. 25PCh. 14.23 - Which one of the C5H8 isomers shown has its max at...Ch. 14.24 - Knowing what to look for with respect to isotopic...Ch. 14.24 - The base peak appears at m/z105 for one of the...Ch. 14.24 - Mass spectra of 1-bromo-4-propylbenzene and...Ch. 14.25 - Prob. 30PCh. 14 - Each of the following compounds is characterized...Ch. 14 - Deduce the structure of each of the following...Ch. 14 - From among the isomeric compounds of molecular...Ch. 14 - The H1NMR spectrum of fluorene has signals at 3.8...Ch. 14 - Prob. 35PCh. 14 - H1NMR spectra of four isomeric alcohols with...Ch. 14 - Prob. 37PCh. 14 - We noted in Section 14.13 that an NMR spectrum is...Ch. 14 - Identify each of the C4H10O isomers on the basis...Ch. 14 - A compound (C3H7ClO2) exhibited three peaks in its...Ch. 14 - Label nonequivalent carbons in the following...Ch. 14 - Compounds A and B are isomers of molecular formula...Ch. 14 - C13 NMR spectra for four isomeric alkyl bromides...Ch. 14 - Prob. 44PCh. 14 - Prob. 45PCh. 14 - Identify the C3H5Br isomers on the basis of the...Ch. 14 - Prob. 47PCh. 14 - A compound (C8H10O) has the IR and H1NMR spectra...Ch. 14 - Deduce the structure of a compound having the...Ch. 14 - Figure 14.53 presents IR, H1NMR, C13NMR and mass...Ch. 14 - H1NMR, C13NMR, IR, and mass spectra are shown for...Ch. 14 - 1H NMR and IR spectra for a compound with the...Ch. 14 - FriedelCraftsalkylation of benzene with...Ch. 14 - Prob. 54DSPCh. 14 - Prob. 55DSPCh. 14 - Prob. 56DSPCh. 14 - Prob. 57DSPCh. 14 - Prob. 58DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Below are two molecules, cyclohexylamine (A) and aniline (B). When analyzed by Infrared Spectroscopy (IR) two different frequencies are observed for the C-N absorption band. Which molecule will have the lower wavenumber absorption band for the C-N bond. Explain why in 35 words or less. „NH2 NH2 А Barrow_forwardYou have the following solutions, A-E. Which solution has the highest absorbance value at 280 nm? Solution A: 1.0 mM Gly-Ser Solution B: 10 mM Gly-Ser Solution C: 1.0 mM Phe-Ser Solution D: 1.0 mM Tyr-Ser Solution E: 1.0 mM Trp-Ser Solution C Solution E Solution B Solution D Solution Aarrow_forwardChemistry CN NC OFF what is the wavelength of the HOMO- LUMO transition in this polyene dyes?arrow_forward
- Which of the following compounds absorbs at the Longest Wavelength (Highest λmax)?arrow_forwardCalculate the IHD of C7H6XNO and identify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.arrow_forward2. What is the concentration (mM) of a solution of tryptophan if the absorbance at 280 nm is 0.850? The cell path length is 1.0 cm and the extinction coefficient is 5500 M-1 cm-1arrow_forward
- 3. Find the position of the absorption peak of a low-resolution spectrum of 12C180arrow_forwardWhy were the absorbance measurements recorded at lambda max (504 nm)? Question 1 options: at lambda max impurities in the sample do not absorb any light Because the allura red dye absorbs red color at 504 nm at lambda max the absorbance is the most sensitive to dye concentration at lambda max the absorbance is the least sensitive to dye concentrationarrow_forwardAssign the correct constitutional isomer to this spectrum (C5H12O)arrow_forward
- 22. Which of the following compounds gives an infrared Spectrum with peaks at 3300cm (sharp Peak) and 2150 cm (sharp Peak) ? H LH ₂ CH ₂ C = CH CH3C=CCH 3 2 1 HCCH H₂C A) I B)2 c) 3 04 CAZ H 9 CH₂arrow_forwardA 3.7 × 10−4 M solution of compound X has an absorbance of 0.5342 when measured at 740 nm. Assuming a pathlength of 1 cm, calculate compound X’s molar absorptivity.arrow_forward5. A solution of 1.1 g/mL of a pure R enantiomer in a 1.0 dm polarimeter rotates plane polarized light by +23.4°. What is the rotation observed on this solution in a 4 dm polarimeter?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY