
(a)
Interpretation:
The total number of nitrogen atoms present in the
Concept Introduction:
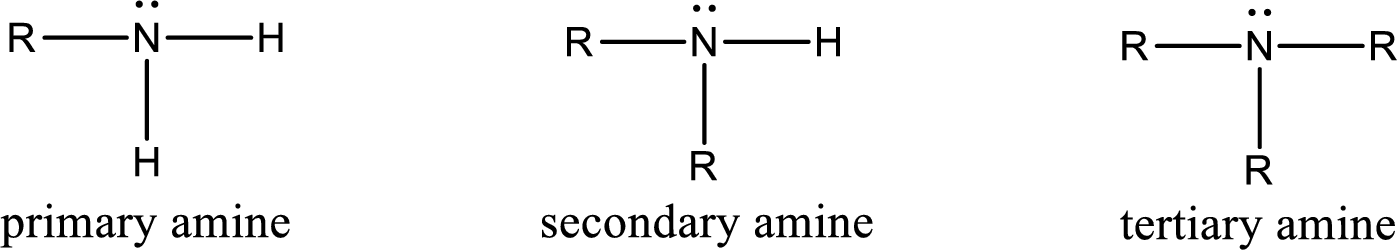
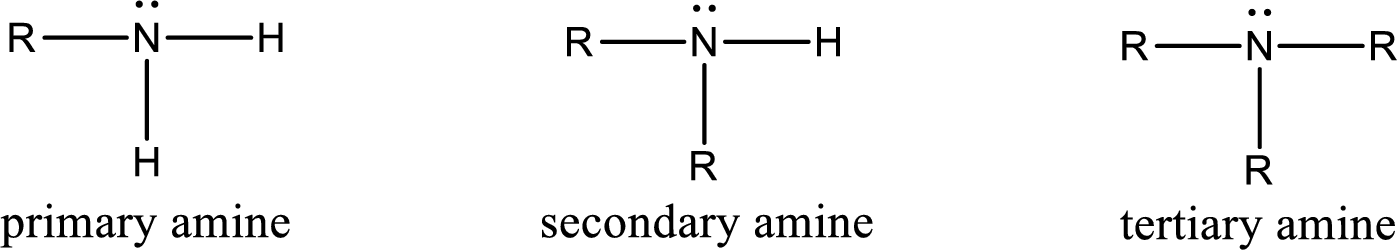
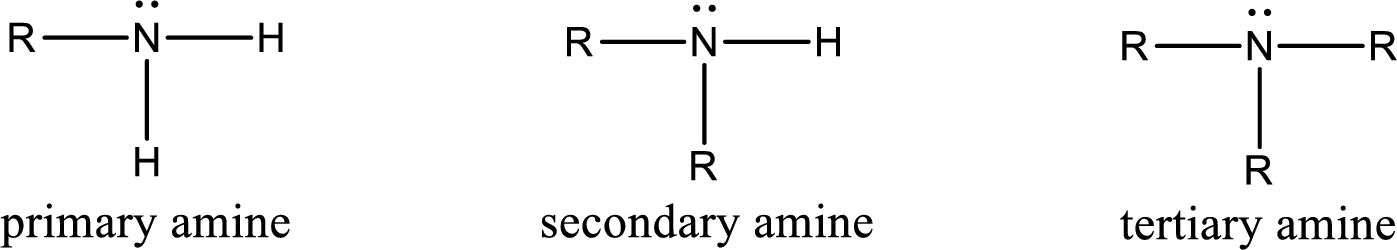
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the
(b)
Interpretation:
The total number of nitrogen atoms present in the functional group of secondary amine has to be given.
Concept Introduction:
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
(c)
Interpretation:
The total number of nitrogen atoms present in the functional group of tertiary amine has to be given.
Concept Introduction:
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
General, Organic, and Biological Chemistry
- Classify each of the molecules below. 1° amine 1° amine 1° amine 2° amine 2° amine 3° amine 3° amine not an amine at all not an amine at all 2° amine 3° amine not an amine at allarrow_forwardCyclic amides are called which of the following? a. lactams b. lactanes c. lactones d. lactinesarrow_forwardHow many R groups are present in each of the following types of amides? A. Unsubstituted amide B. Disubstituted amide C. Secondary amidearrow_forward
- Draw condensed and skeletal structures for each of the following amines a. 2-methyl-N-propyl-1-propanamine b. N-ethylethanamine c. 5-methyl-1-hexanamine d. methyldipropylaminee. e. N,N-dimethyl-3-pentanamine f. cyclohexylethylmethylaminearrow_forward10. Which of the following molecules is a secondary amine? NH2 NH2 П III IV a. I and II b. I and III c. II and III d. III and IVarrow_forwardIV: Classify each of the following amines as primary, secondary or tertiary amines. 1. Dibutylamine 2. Cyclohexanamine 3. m-Chloroaniline 4. 2-Methyl-2-heptanamine 5. Tripentylaminearrow_forward
- Draw a condensed structural formula for each of the following amine?A. 2-Methyl-3-ethyl-1-hexanamine B. 1,3-Pentanediaminearrow_forwardWhich of the following types of compounds does not contain a carbonyl group? O A. primary amine O B. acid anhydride O C. ethyl ester O D. primary amidearrow_forwardwhat is the difference between an amine and an amide?arrow_forward
- Which of the following statements is true for an amine if "N-" is part of the IUPAC name? a. The compound is a primary amine. b. The molecule is contains a nitrogen atom attached to carbon number one. c. The compound is a secondary amine. d. The compound is a tertiary amine.arrow_forward13. Write the chemical reaction for the base hydrolysis of each amide sample. Include the condensed structural formulas of the reactants and products. ACETAMIDE BENZAMIDEarrow_forwardHow many carbon-nitrogen bonds are present in each of the following types of amines? a. primary amine b. secondary amine c. tertiary amine 17-4arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





