
(a)
Interpretation: The IUPAC name of the following compound should be determined:
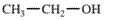
Concept Introduction:
In order to give the IUPAC name to the alcohol following steps are followed:
1. The parent (longest)
2. The ending of the parent chain from alkane (-e) is changed to -ol and the number is used to locate the -OH group of alcohol.
3. Name should be written in alphabetical order and numbering should be done in such a way that hydroxy group gets lowest number.
4. Configuration should be specified is there exist any cis-trans isomerism.
For number of carbons atoms in chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
(b)
Interpretation: The IUPAC name of the following compound should be determined:
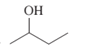
Concept Introduction:
In order to give the IUPAC name to the alcohol following steps are followed:
1. The parent (longest) alkane chain is identified.
2. The ending of the parent chain from alkane (-e) is changed to -ol and the number is used to locate the -OH group of alcohol.
3. Name should be written in alphabetical order and numbering should be done in such a way that hydroxy group gets lowest number.
4. Configuration should be specified is there exist any cis-trans isomerism.
For number of carbons atoms in chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
(c)
Interpretation: The IUPAC name of the following compound should be determined:
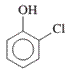
Concept Introduction:
In order to give the IUPAC name to the alcohol following steps are followed:
1. The parent (longest) alkane chain is identified.
2. The ending of the parent chain from alkane (-e) is changed to -ol and the number is used to locate the -OH group of alcohol.
3. Name should be written in alphabetical order and numbering should be done in such a way that hydroxy group gets lowest number.
4. Configuration should be specified is there exist any cis-trans isomerism.
For number of carbons atoms in chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Basic Chemistry
- What is the IUPAC name of the compound below? H. O 3.7-dimethyl-6-one-4-octynal O 2.6-dimethyl-3-one-4-octynal 3,7-dimethyl-6-oxo-4-octynal 2,6-dimethyl-3-oxo-4-octynalarrow_forwardWhat is the IUPAC name of the compound below? to OH Multiple Choice 2,2-dimethyl-1-propanol 2,2-dimethyl-4-butanol 2,2-dimethyl-3-propanol 3,3-dimethyl-1-butanolarrow_forward16. The correct IUPAC name for the compound shown below is? OHarrow_forward
- Draw the condensed structural formula for the ether produced by each of the following reactions: H+ а. 2СH3 — ОН Heat H* b. 2CH3-CH2–CH2-OH Heatarrow_forwardWhat is the IUPAC name of the following compound? CH3 CH3-C=C-CH,-CH-CH3 Select one: A. 5-methyl-3-hexyne B. 2-methyl-4-hexyne C. 6-methyl-3-hexyne D. 5-methyl-2-hexyne E. 4-methyl-2-hexynearrow_forwardWhat is the IUPAC name of the molecule below? CH3 CH;CH,CHCH CHCH CH C-OH Select one: a. octanoic acid O b. 6-ethyl-4-methylhexanoic acid O c. 4,6-dimethyloctanoic acid O d. 3,5-dimethyloctanoic acidarrow_forward
- What is the IUPAC name for this compound? CH3 0 CH3-CH-CH2-C-OH O pentanoic acid 1 02-methylbutanoic acid O2-methyl-4-butanoic acid 03-methylbutanoic acid O2-methyl butyric acid Question 44 0 Which of the following ketones is the most soluble in water? 0 1 CH3-CH2-C-CH2-CH3 0 CH3-CH-C-CH3 CH3arrow_forwardNomenclature for Ketones (Section 4-5) 31 Using IUPAC nomenclature, name each of the foll. . ketones. a. CH3-CH2-C-CH3 b. CH3-CH-CH--C-CH-CH3 CH3 CH3 CH3arrow_forwardThe name of CH3-CH C CH-CH-CH CH-CH3 is Select one: a. 2, 3, 6- octatriene b. 3, 5, 6- octatriene C. 2, 5, 6- octatriene d. 2, 3, 5 - octatrienearrow_forward
- The correct IUPAC name for this compound is: O a. 4-oxa-1-cyclohexanol O b. 4-hydroxyoxacyclohexene O c. 4-oxa-1-cyclohexenol O d. 4-oxacyclohexane-1-ol OHarrow_forwardWhat is the IUPAC name of the following compound? OH A. 4-methyl-5-penten-1-ol B. 4-methyl-4-penen-1-ol O C. 5-hydroxy-2-methyl-1-pentene D. 2-methyl-1-penten-5-olarrow_forward614 Give the IUPAC name for each of the following 5-14 carboxylic acids. a. CH3-CH2-CH2-CH2-C-OH CH3 CH3 bilk b. CH—CH-СН,—С—ОН CH3 CH3 O c. CH3-CH-CH-C-OH CH-CH—С-ОН d. CH3-CH,—СH,—СООНarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





