
Concept explainers
(a)
Interpretation:
Dominant resonance hybrid structure among below two Lewis structures have to be determined.

Concept Introduction:
Resonance hybrid structures are set of two or more Lewis structures that illustrate electronic bonding through fractional bonds or fractional charges. Dominant resonance structure possesses the rules as follows:
1. Complete octet of each atom.
2. Smallest number of formal charges.
3. Most electronegative atom should possess negative charge and most electropositive atom should possess positive charge.
The formal charge is the charge assigned on each atom irrespective of its electronegativity so that electrons are equally shared between the atoms. The formal charge on each atom in the Lewis structure can be calculated from the equation written as follows:
Here,
(b)
Interpretation:
Dominant resonance hybrid structure among below two Lewis structures have to be determined.
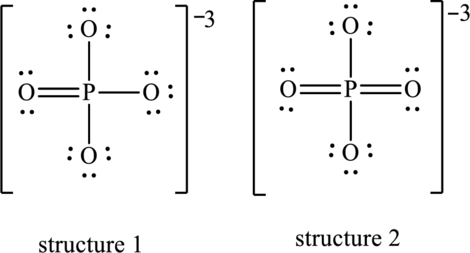
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- The cyclohexane carboxylate anion has a Lewis structure Pushing a pair of unshared electrons away from the negatively charged oxygen atom and, at the same time, pushing a pair of pi electrons toward the other oxygen will generate a second resonance structure. Thus,arrow_forwardValine is an amino acid with this Lewis structure: Write the Lewis structure for the zwitterion form of valine.arrow_forwardIn molecules of the type X-O-H, as the electronegativity of X increases, the acid strength increases. In addition, if the electronegativity of X is a very small value, the molecule acts as a base. Explain these observations and provide examples.arrow_forward
- Complete the following Lewis structures for the CICN molecule by adding dots for unshared valence electrons and indicating formal charges. Evaluate the importance of each structure as a contributor to a resonance hybrid. Cl – C = N, Cl = C – N, Cl = C = Narrow_forwardComplete the following Lewis structures for the HONS molecule by adding dots for unshared valence electrons and indicating formal charges. Evaluate the importance of each structure as a contributor to a resonance hybrid. H – 0 – N = S, H–0 = N – Sarrow_forwardSulfuric acid is the industrial chemical produced in greatest quantity worldwide. About 90 billion pounds are produced each year in the United States alone. Write the Lewis structure for sulfuric acid, H2SO4, which has twooxygen atoms and two OH groups bonded to the sulfur.arrow_forward
- Which of the following statements concerning the structures below is/are true? You can select more than one, or none, of these statements. N = A N: Z: = N B = Z: :N=N- The total charge on this species is -1. Structure B is the least important structure. For each N in structure B, the formal charge is zero. Structures A and B are equivalent resonance structures. Structures A, B, and C are equivalent resonance structures. Structures A and C are equivalent resonance structures. In structure A, the N atom on the left has a formal charge of zero. C N:arrow_forwardWrite the Lewis structure for the diatomic molecule P2, an unstable form of phosphorus found in hightemperature phosphorus vapor.arrow_forwardA 0.167-g sample of an unknown compound contains 0.00278 mol of the compound. Elemental analysis of the compound gives the following percentages by mass: 40.00% C; 6.71% H; 53.29% O. Determine the molecular formula, molar mass, and Lewis structure of the unknown compound.arrow_forward
- Write all alternative (total of six) Lewis structures of CISO*. Calculate formal charges on each atom in each structure. Apply formal charge rules to select the most plausible structure among all alternative structures. Justify your selection.arrow_forwardA 0.167-g sample of an unknown acid requires 27.8 mL of 0.100 M NaOH to titrate to the equivalence point. Elemental analysis of the acid gives the following percentages by mass: 40.00% C, 6.71% H, 53.29% O. Determine the molecular formula, molar mass, and Lewis structure of the unknown acid.arrow_forwardWrite the Lewis structure in the 2nd column and molecular geometry in the 3rd column.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



