
Concept explainers
(a)
Interpretation:
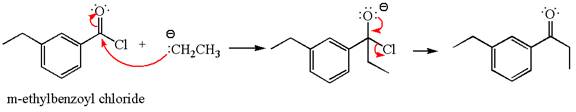
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to corresponding
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
Explanation of Solution
The equation for the reaction of
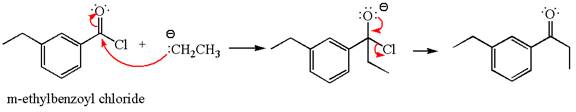
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(b)
Interpretation:
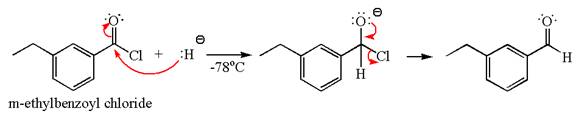
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to an
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
Explanation of Solution
The equation for the reaction of
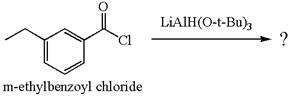

The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(c)
Interpretation:
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to a primary alcohol by reacting with
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
Explanation of Solution
The equation for the reaction of
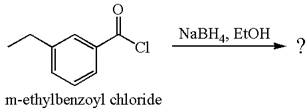
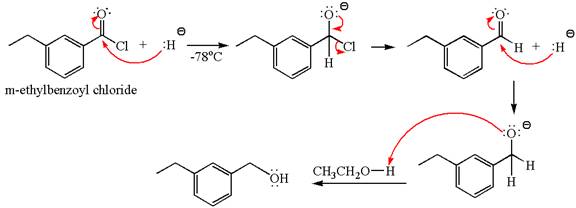
The product with detailed mechanism for the given reaction is drawn based on the reactivity of
(d)
Interpretation:
The product with detailed mechanism for the reaction between
Concept introduction:
An acid chloride can be converted to a tertiary alcohol by reacting it with
Answer to Problem 20.45P
The product with detailed mechanism for the reaction between
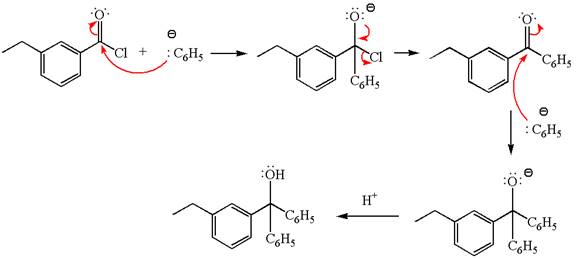
Explanation of Solution
The equation for the reaction of
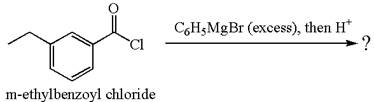

The product with detailed mechanism for the given reaction is drawn based on the reactivity of
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- 22. Provide the following: 1) the major organic product of each reaction shown, and ii) the specific name of the reaction in the first step. aniline AICI (if o,p-substituted products are possible, only provide the major para-substituted product, otherwise, only provide the meta-substituted product) 12, HNO (if o,p-substituted products are possible, only provide the major para-substituted product, otherwise, only provide the meta-substituted product) end a chatarrow_forward1// In a set of the given reactions, acetic acid yielded a product C C6 H6 C HM9B CH;COOH + PCl, → A anh. AlCl ether What would be product C? 2// When propionic acid is treated with aqueous sodium bicarbonate formed? 3// When CH3COOH reacts with CH3-Mg-X formed? 4// What is the product in the given reaction when benzyl chloride reacts with benzen?arrow_forwardDetermine whether each of the following syntheses requires a reaction that alters the carbon skeleton. (a) (b) OH (c) (d) OCH,CH3 CH3 (e) OH (f) H,CO он H;CO. (g) H3CO, (h) H3CO. NO2 NO2arrow_forward
- Draw the products formed when benzoyl chloride (C,H;COCI) is treated with each nucleophile: (a) H20, pyridine; (b) CH;CO0"; (c) NH3 (excess); (d) (CHaNH (excess).arrow_forward10. Compound X (C4H9Br) reacts by heating with NaOH in H2O to form Y. The compound Y then undergoes acid catalysed hydration by H2SO, in 180°C to form 2-methyl prop-1-ene. (e) Determine the structure of X and Y. (f) Predict a MAJOR product when compound Y reacts with H2SO4 in 140 C. (g) Draw a structural isomer of X. Name the isomer using IUPAC nomenclature. (h) Describe a chemical test to distinguish between compound Y and 1-butanol.arrow_forwardDraw the product formed when CH3CH2C=CH is treated with each of the following sets of reagents: (a) H2O, H2SO4, HgSO4; and (b) R2BH, followed by H2O2, HO−.arrow_forward
- Name the structure. Heating compound A in the presence of H2SO4 results in which product?arrow_forward4. identify the structure of A and B in the following synthetic scheme: Write out complete reactions for each step, showing the structure of all reactants and products. (a) cyclohexanol +Na2Cr2O7/H2SO4, H2O -------> A (b) A + Et2NH/H2SO4 ----->Barrow_forwardDevise at least three different methods to prepare N-methylbenzylamine (PhCH2NHCH3) from benzene, any one-carbon organic compounds, and any required reagents.arrow_forward
- (a) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers that are not tautomers, but contain a C=C and an OH group. 2-methylcyclohexanonearrow_forwardGive the major product of the following reaction. CH;CH,OH CIarrow_forwardβ-Vetivone is isolated from vetiver, a perennial grass that yields a variety of compounds used in traditional eastern medicine, pest control, and fragrance. In one synthesis, ketone A is converted to β-vetivone by a two-step process:Michael reaction, followed by intramolecular aldol reaction. (a) What Michael acceptor is needed for the conjugate addition? (See Problem 23.61 for another method to form the bicyclic ring system of β-vetivone.) (b) Draw a stepwise mechanism for the aldol reaction, which forms the six-membered ring.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





