
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 22.50P
Following is the structure of miconazole, the active antifungal agent in a number of over-the-counter preparations, including Monistat, that are used to treat vaginal yeast infections. One of the compounds needed for the synthesis of miconazole is the trichloro derivative of toluene shown on its right.
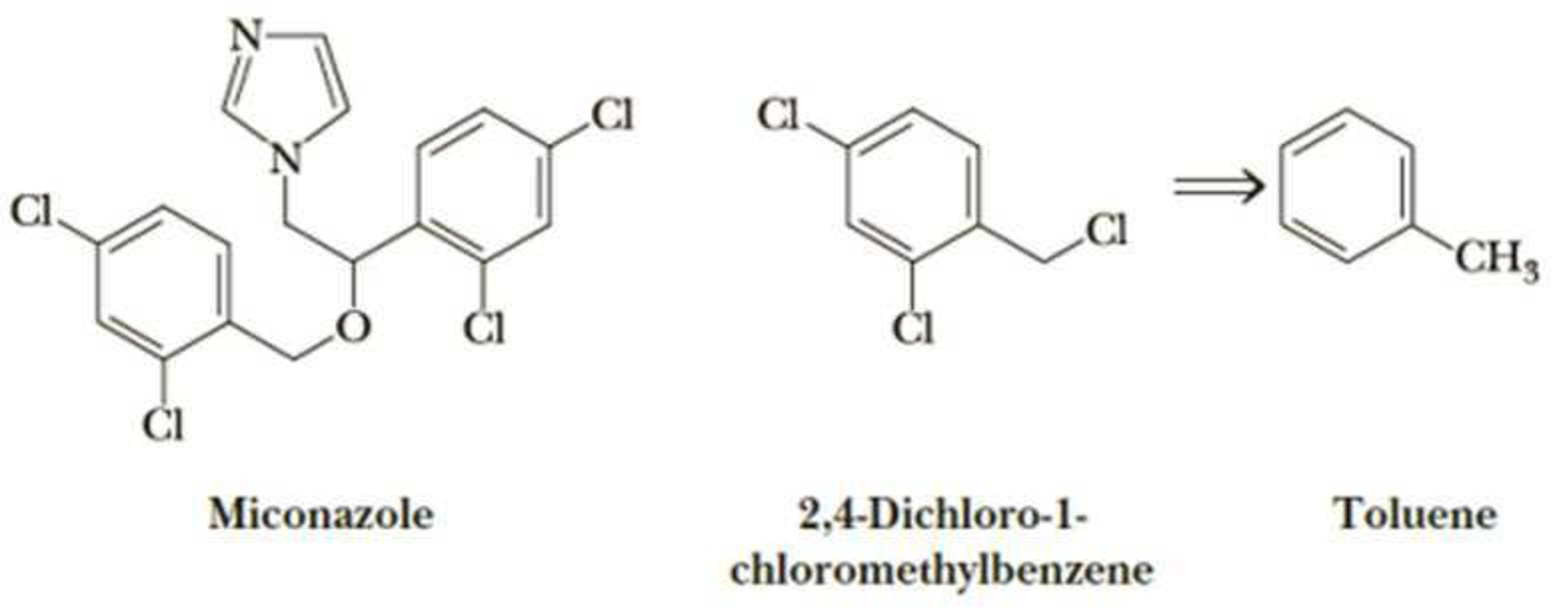
- (a) Show how this derivative can be synthesized from toluene.
- (b) How many stereoisomers are possible for miconazole?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) Draw the structure of a compound of molecular formula C6H10 that reacts with H2 in the presence of Pd-C but does not react with H2 in the presence of Lindlar catalyst. (b) Draw the structure of a compound of molecular formula C6H10 that reacts with H2 when either catalyst is present.
The hydrocarbon fluorene was treated with potassium t-butoxide in an acid-base reaction, giving the fluorenide anion and t-butyl alcohol. (a) Which way does the equilibrium lie, and by how much?
b) What is the proportion of the fluorenide anion to fluorene?
(c) Why is fluorene so highly acidic, considering the pKa of an average alkane is above 50?
Resveratrol is an antioxidant found in the skin of red grapes. Its anticancer, anti-inflammatory, and various cardiovascular effects are under active investigation. (a) Draw all resonance structures for the radical that results from homolysis of the OH bond shown in red. (b) Explain why homolysis of this OH bond is preferred to homolysis of either OH bond in the other benzene ring.
Chapter 22 Solutions
Organic Chemistry
Ch. 22.1 - Prob. 22.1PCh. 22.1 - Write a structural formula for the product from...Ch. 22.1 - Prob. 22.3PCh. 22.2 - Prob. 22.4PCh. 22.2 - Predict the major produce(s) of each electrophilic...Ch. 22.3 - In SN2 reactions of haloalkanes, the order of...Ch. 22 - Prob. 22.8PCh. 22 - Prob. 22.9PCh. 22 - Addition of m-xylene to the strongly acidic...Ch. 22 - Addition of tert-butylbenzene to the strongly...
Ch. 22 - What product do you predict from the reaction of...Ch. 22 - Other groups besides H+ can act as leaving groups...Ch. 22 - Prob. 22.14PCh. 22 - Prob. 22.15PCh. 22 - Prob. 22.16PCh. 22 - Prob. 22.17PCh. 22 - Suggest a reason why the nitroso group, N=O, is...Ch. 22 - Prob. 22.19PCh. 22 - Prob. 22.20PCh. 22 - The following molecules each contain two aromatic...Ch. 22 - Prob. 22.22PCh. 22 - Prob. 22.23PCh. 22 - The insecticide DDT is prepared by the following...Ch. 22 - Prob. 22.25PCh. 22 - Prob. 22.26PCh. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Prob. 22.32PCh. 22 - Show how to prepare each compound from...Ch. 22 - Prob. 22.34PCh. 22 - Show reagents and conditions to bring about the...Ch. 22 - Prob. 22.36PCh. 22 - Propose a synthesis for each compound from...Ch. 22 - The first widely used herbicide for the control of...Ch. 22 - The first widely used herbicide for the control of...Ch. 22 - Prob. 22.40PCh. 22 - Prob. 22.41PCh. 22 - Prob. 22.42PCh. 22 - Prob. 22.43PCh. 22 - Cancer of the prostate is the second leading cause...Ch. 22 - Prob. 22.45PCh. 22 - Prob. 22.46PCh. 22 - Prob. 22.47PCh. 22 - When certain aromatic compounds are treated with...Ch. 22 - Prob. 22.49PCh. 22 - Following is the structure of miconazole, the...Ch. 22 - Prob. 22.51PCh. 22 - Prob. 22.52PCh. 22 - Prob. 22.53PCh. 22 - Show how the antidepressant venlafaxine (Effexor)...Ch. 22 - Prob. 22.57PCh. 22 - Given this retrosynthetic analysis, propose a...Ch. 22 - Prob. 22.59PCh. 22 - Prob. 22.60PCh. 22 - Prob. 22.61PCh. 22 - A newer generation of antipsychotics, among them...Ch. 22 - Prob. 22.63P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Resveratrol is an antioxidant found in the skin of red grapes. Its anticancer, anti-inammatory, and various cardiovascular effects are under active investigation. (a) Draw all resonance structures for the radical that results from homolysis of the OH bond shown in red. (b) Explain why homolysis of this OH bond is preferred to homolysis of either OH bond in the other benzene ring.arrow_forwardThe bicyclic heterocycles quinoline and indole undergo electrophilic aromatic substitution to give the products shown. (a) Explain why electrophilic substitution occurs on the ring without the N atom for quinoline, but occurs on the ring with the N atom in indole. (b) Explain why electrophilic substitution occurs more readily at C8 than C7 in quinoline. (c) Explain whyelectrophilic substitution occurs more readily at C3 rather than C2 of indole.arrow_forward(a) Which of the following phenols is the least acidic? (b) Which of the following phenols is the most acidic?arrow_forward
- 4) Aromatic compounds are among the most abundant and versatile in nature. From a synthetic point of view, these compounds, despite their stabilities, are quite useful and can undergo reactions under special conditions and by specific mechanisms, such as the Electrophilic Aromatic Substitution (SAE) and the Nucleophilic Aromatic Substitution (SNAr). Based on this, please answer the following items: (a) What are the possible isomeric products for the following reaction? Which structure, A or B, do you expect to predominate? Justify your choice. Write down the detailed mechanism of formation of compounds A and B. What would be the bromination product of each (compounds C and D)?arrow_forward11:43 Q1. (a) (c) (d) (b) Two stereoisomers of but-2-ene are formed when 2-bromobutane reacts with ethanolic potassium hydroxide. (i) Explain what is meant by the term stereoisomers. Library Name and outline a mechanism for the reaction of 2-bromo-2-methylpropane with ethanolic potassium hydroxide to form the alkene 2-methylpropene, (CH3)2C=CH₂ Name of mechanism Mechanism (ii) Draw the structures and give the names of the two stereoisomers of but-2-ene. Stereoisomer 1 Name (iii) Name this type of stereoisomerism. Select Name Stereoisomer 2 When 2-bromo-2-methylpropane reacts with aqueous potassium hydroxide, 2-methylpropan-2-ol is formed as shown by the following equation. CH3 H₂C-C-CH3 + KOH Br Page 2 of 14 CH3 H3C-C-CH3 + KBr ОН State the role of the hydroxide ions in this reaction. Write an equation for the reaction that occurs when CH3CH₂CH₂CH₂Br reacts with an excess of ammonia. Name the organic product of this reaction. Equation Name of product 9,284 Photos, 1,166 Videos For You…arrow_forward(a) Which of the following will NOT produce a carboxylic acid or carboxylate ion? 1-butanol + H2CrO4 2-butene + O3/H2O2 butanal + PCC butanal + H2CrO4 (b) Which of the following will NOT produce a carboxylic acid or carboxylate ion? 1-butanol + H2CrO4 2-butene + O3/H2O2 butanal + PCC butanal + H2CrO4arrow_forward
- Compound A has molecular formula C7H15B.. Treatment of compound A with sodium ethoxide yields only one elimination product (compound B) and no substitution products. When compound B is treated with dilute sulfuric acid, compound C is obtained, which has molecular formula C7H160. Draw the structures of compounds A, B, and C.arrow_forwardRank the compounds in each group in order of increasing reactivity in electrophilic aromatic substitution: (a) C6H6, C6H5Cl, C6H5CHO, C6H5OCH3; (b) C6H5CH3, C6H5NH2, C6H5CH2NH2, C6H5CONH2.arrow_forwardThe bicyclic heterocycles quinoline and indole undergo electrophilic aromatic substitution to give the products shown. (a) Explain why electrophilic substitution occurs on the ring without the N atom for quinoline, but occurs on the ring with the N atom in indole. (b) Explain why electrophilic substitution occurs more readily at C8 than C7 in quinoline. (c) Explain why electrophilic substitution occurs more readily at C3 rather than C2 of indole.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY