
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23, Problem 32P
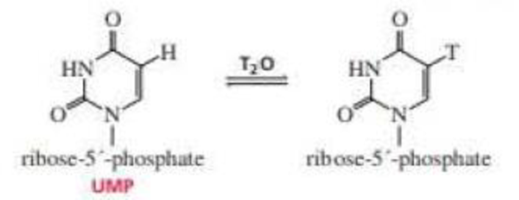
When UMP is dissolved in T2O, exchange of T for H occurs at the 5-position. Propose a mechanism for this exchange. (T=3H; see Problem 31)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When UMP is dissolved in T2O, exchange of T for H occurs at the 5-position. Propose a mechanism for this exchange.
Fill in the missing structures and add curved arrows to show the mechanism of glycoside formation
below.
Step 1: Oxonium ion formation
НО
НО
НО
ОН
НО-
HO OH
D-glucose
Step 2: Alcohol addition
OH
+
H-OH2
HO
H-OR
or
H-OR
НО
НО
OH
HO
oxonium ion
:ÖH2
:ÖH2
НО
HO
НО
+ H2O
НО
ОН
HO
a B-D-glycoside
OH
НО
OR
OR
an a-D-glycoside
Provide an electron pushing mechanism for the conversion shown below. Abbreviate the enzyme catalyst
active site groups (BH* and B:) as needed. Which purine base is used?
NH₂
Enz
H₂O
'N
NH
Chapter 23 Solutions
Organic Chemistry (8th Edition)
Ch. 23.1 - Prob. 2PCh. 23.1 - Prob. 3PCh. 23.2 - How many conjugated double bonds are there in a....Ch. 23.2 - Instead of adding to the 4a position and...Ch. 23.2 - Prob. 7PCh. 23.3 - Prob. 8PCh. 23.3 - Acetolactate synthase is another TPP-requiring...Ch. 23.3 - Acetolactate synthase transfers the acyl group of...Ch. 23.3 - Prob. 12PCh. 23.5 - Which compound is more easily decarboxylated?
Ch. 23.5 - Prob. 14PCh. 23.5 - Explain why the ability of PLP to catalyze an...Ch. 23.5 - Explain why the ability of PLP to catalyze an...Ch. 23.5 - The enzyme that catalyzes the C C bond cleavage...Ch. 23.5 - Propose a mechanism for the ,-elimination reaction...Ch. 23.6 - Ethanolamine ammonia lyase, a coenzyme...Ch. 23.6 - Prob. 20PCh. 23.7 - How do the structure of tetrahydrofolate and...Ch. 23.7 - What is the source of the methyl group in...Ch. 23.8 - Thiols such as ethanethiol and propanethiol can be...Ch. 23 - How does the metal ion in carboxypeptidase A...Ch. 23 - Prob. 24PCh. 23 - Prob. 25PCh. 23 - For each of the following reactions, name both the...Ch. 23 - Prob. 27PCh. 23 - When transaminated, the three branched-chain amino...Ch. 23 - What acyl groups have we seen transferred by...Ch. 23 - Propose a mechanism for the following reaction:Ch. 23 - Draw the products of the following reaction, where...Ch. 23 - When UMP is dissolved in T2O, exchange of T for H...Ch. 23 - Dehydratase is a PLP-requiring enzyme that...Ch. 23 - In addition to the reaction mentioned in Section...Ch. 23 - PLP can catalyze both ,-elimination reactions...Ch. 23 - The glycine cleavage system is a group of four...Ch. 23 - Prob. 37PCh. 23 - FADH2 reduces , -unsaturated thioesters to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M- 1s - 1. In the presence of Co2 +, the rate constant is 1.5 * 106 M- 1s - 1. What rate enhancement does the catalyst provide?arrow_forwardc) Study the following reaction scheme for the preparation of a natural glycoside (compound 7) and answer the questions that follow: OH OAc НО excess Ac,O AcO OH pyridine, 0°C AcO- OAc НО OAc Compound 3 Compound 4 HBr OCH3 OAc OAc AcO AcO AcO AcO- OAc OAc Compound 6 Compound 5 Br NaOH, H,O OCH3 OH HO НО. Compound 7 Write a mechanism for the reaction of any one of the hydroxyl groups of compound 3 in the conversion to compound 4. i.arrow_forwardThe enzyme that catalyzes the Ca ¬ Cb bond cleavage reaction that converts serine to glycine removes thesubstituent (R) bonded to the a-carbon in the first step of the reaction. Starting with PLP bound to serine inan imine linkage, propose a mechanism for this reaction. (Hint: The first step involves removal of the protonfrom serine’s OH group.)arrow_forward
- One of the later steps in glucose biosynthesis is the isomerization of fructose 6-phosphate to glucose 6-phosphate. Propose a mechanism, using acid or base catalysis as needed.arrow_forwardPlease draw by hand. Triosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme's catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a base and an acid catalyst, respectively. Propose a mechanism for the reaction. ОН 2-03Р0 ОН dihydroxyacetone phosphate triosephosphate isomerase 2-03РО. H glyceraldehyde-3-phosphate FYI Glu is glutamic acid and his is histadinearrow_forwardACTIVITY-4. Refer to the structures below. Draw the Haworth formula (pyranose) -OH но H- FOH FOH- но но H- H FOH H FOH- H- CH2OH CH2OH Alpha-D- glucopyranose Beta-D-glucopyranose (Haworth) Beta-D-glucopyranose Alpha-D- glucopyranose (Haworth) ACTIVITY-5. Draw the Haworth formula for D-mannose and D-galactose. Label the alpha and beta anomer. CYCLIC FORMS OF FRUCTOSE D-fructose, a ketohexose sugar, can be presented using furanose and pyranose. Cyclic Fisher (furanose) 1 2OH Open chain Fisher Haworth formula (furanose) 1 CH2OH HOH,C- 1 2 F0 НО 3_H. ÇH2OH ÇH2OH но з—н 4 H- H но 2 H-4 HO- H- 5. OH 13 H-5 FOH 6 CH,OH 14 OH H. 6 CH2OH D-fructose Alpha form Alpha form Cyclic Fisher (pyranose) Haworth formula (pyranose) 1 HOH2C-2 3. 1 HO- CH2OH Но H. 5 он 2 H-4 HO- Но 4 OH 3 H-5 FHO- OH H-6 H Alpha form of D-fructose Alpha form of D-fructose ACTIVITY-6. Draw the Fisher and Haworth formula for beta-D-fructose (furanose and pyranose form). Label the beta furanose and beta pyranose.arrow_forward
- 3. Propose a mechanism for the conversion from ribulose5-5phosphate to ribose-5-phosphate using the arrow formalism. This is a reaction in the pentose phosphate pathway. CH2OH с-н Ribose 5-Phosphate Isomerase c=0 H-C-OH Н-с-ОН Н-с-ОН Н-с-он Н-с-ОН H2C-OPO,2 H2C-OPO, Ribulose-5-Phosphate Ribose-5-Phosphatearrow_forwardThe disaccharide lactulose consists of a d-galactopyranose subunit and a d-fructofuranose subunit joined by a β-1,4-glycosidic linkage. After treatmentof lactulose with 1. excess CH3I/Ag2O, 2. HCl/H2O, the d-galactopyranose subunit was found to have one nonmethylated OH group, whereas thed-fructofuranose subunit had two. Draw the structure of α-lactulose.arrow_forwardlysozyme catalyzes the hydrolysis of a carbohydrate linkage in part of the bacterial wall. The active site of the lysozyme contains aspartate and glutamate side chains. 1. lysozyme catalyzes this reaction with a catalytic Zn 2+ ion. propose a mechanism.arrow_forward
- 29. The major product in the following reaction sequence is (i) mCPBA, NaHCO3 (ii) NaOH, H2O MeO (iii) I2, KI, NAHCO3, H20arrow_forwardShown here is a/anglycosidic linkage. CH₂OH CH₂OH I OH Н OH Н O Alpha Beta OL- O D- О Н I ОН HO OH -О Н Н H OH OHarrow_forwarddoes structure E represent fructofuranose? explainarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY