
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 34P
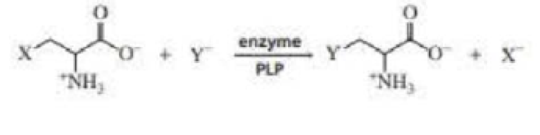
In addition to the reaction mentioned in Section 23.5. PLP can catalyze β-substitution reactions. Propose a mechanism for the following PLP-catalyzed β-substitution reaction:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are the similarities between β oxidations of heptadecanoic acids (C 17:0), and cis-Δ9-heptadencanoic acids (C 17:1).
The first step in the catabolism of most amino acids is the removal of the nitrogen atom by transfer to an a-keto acid, a reaction
catalyzed by an enzyme called a transaminase. The a-keto acid acceptor is often a-ketoglutarate. Modify the structures in the
product to show the products of the transamination of cysteine. Be sure to show functional groups with the charge and number of
attached hydrogen atoms appropriate for pH 7.4.
transaminase
+
O=C
H₂N-CH
+
CH₂
CH₂
CH₂
SH
Incorrect
H₂N
||
CH
|
CH₂
|
CH₂
I
||
O
||
n
|
CH₂
T
SH
Please draw by hand. Triosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme's catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a base and an acid catalyst, respectively. Propose a mechanism for the reaction.
ОН
2-03Р0
ОН
dihydroxyacetone phosphate
triosephosphate isomerase
2-03РО.
H
glyceraldehyde-3-phosphate
FYI Glu is glutamic acid and his is histadine
Chapter 24 Solutions
Organic Chemistry
Ch. 24.1 - Prob. 2PCh. 24.1 - Prob. 3PCh. 24.2 - How many conjugated double bonds are there in a....Ch. 24.2 - Instead of adding to the 4a position and...Ch. 24.2 - Prob. 7PCh. 24.3 - Prob. 8PCh. 24.3 - Acetolactate synthase is another TPP-requiring...Ch. 24.3 - Acetolactate synthase transfers the acyl group of...Ch. 24.5 - Prob. 12PCh. 24.5 - The enzyme that catalyzes the C C bond cleavage...
Ch. 24.5 - Propose a mechanism for the ,-elimination reaction...Ch. 24.5 - Which compound is more easily decarboxylated?Ch. 24.5 - Explain why the ability of PLP to catalyze an...Ch. 24.5 - Explain why the ability of PLP to catalyze an...Ch. 24.6 - Ethanolamine ammonia lyase, a coenzyme...Ch. 24.6 - Prob. 19PCh. 24.7 - How do the structure of tetrahydrofolate and...Ch. 24.7 - What is the source of the methyl group in...Ch. 24.8 - Thiols such as ethanethiol and propanethiol can be...Ch. 24 - How does the metal ion in carboxypeptidase A...Ch. 24 - Prob. 23PCh. 24 - Prob. 24PCh. 24 - Prob. 25PCh. 24 - S-Adenosylmethionine (SAM) is formed from the...Ch. 24 - Prob. 27PCh. 24 - What acyl groups have we seen transferred by...Ch. 24 - Propose a mechanism for the following reaction:Ch. 24 - When transaminated, the three branched-chain amino...Ch. 24 - Draw the products of the following reaction, where...Ch. 24 - When UMP is dissolved in T2O, exchange of T for H...Ch. 24 - Dehydratase is a PLP-requiring enzyme that...Ch. 24 - In addition to the reaction mentioned in Section...Ch. 24 - PLP can catalyze both ,-elimination reactions...Ch. 24 - The glycine cleavage system is a group of four...Ch. 24 - Prob. 37PCh. 24 - FADH2 reduces , -unsaturated thioesters to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following compounds is a weak electrolyte?arrow_forwardGive the functions of the following ingredients, then name a branded/commercial skin or hair care product where the said material is used. Bisabolol (Alpha Bisabolol) Ceresin Wax Disodium Lauryl Sulfosuccinate Hydroxypropyl Methylcellulosearrow_forward10. Enumerate the PUFA (Polyunsaturated Fatty Acids) and indicate the position of unsaturation? 11. Enumerate the derivatives of steroids and their functions. 12. Compare and contrast the functions lipoproteins. 13. Circle the fatty acid in each pair that has the higher melting temperature. (a) 18:1Δ9 18:2 Δ 9,12 (b) 18:0 18:1 Δ 9 (c) 18:0 16:0 14. Describe the dependence of the melting point of a fatty acid upon (a) chain length and (b) unsaturation; (c) explain these dependencies in molecular terms.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY