
Concept explainers
(a)
Interpretation:
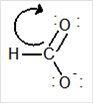
Draw the contributing structure of.
Concept Introduction:
Lewis electron dot symbol is a structure of a molecule that shows the bonding between atoms as well as lone pairs of electrons of atoms also.
Valence electron: The electrons which are present in the outer most energy level are known as valence electron. This can be calculated by the group number of the element. Generally the group number is same as the valence electrons of any elements.
(b)
Interpretation:
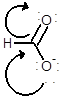
Draw the contributing structure of.
Concept Introduction:
Lewis electron dot symbol is a structure of a molecule that shows the bonding between atoms as well as lone pairs of electrons of atoms also.
Valence electron: The electrons which are present in the outer most energy level are known as valence electron. This can be calculated by the group number of the element. Generally the group number is same as the valence electrons of any elements.
(c)
Interpretation:
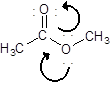
Draw the contributing structure of.
Concept Introduction:
Lewis electron dot symbol is a structure of a molecule that shows the bonding between atoms as well as lone pairs of electrons of atoms also.
Valence electron: The electrons which are present in the outer most energy level are known as valence electron. This can be calculated by the group number of the element. Generally the group number is same as the valence electrons of any elements.
Trending nowThis is a popular solution!

Chapter 3 Solutions
Introduction To General, Organic, And Biochemistry
- 3-26 Table 3-2 shows the following ions of copper: Cu+ and Cu2+. Do these violate the octet rule? Explain.arrow_forwardReview Topice] [References] EXERCISE Drawing Lewis Structures I Clear н с н Complete the Lewis structure by dragging bonds and lone electron pairs to their appropriate positions. Click on an atom to remove an electron pair from that atom or click on a bond to change its order. The structure contains 12 valence electrons. Check Nuxt (4 of 16) Show Approacth Submit Answer Retry Entire Group No more group attempts remainarrow_forward& BE 4 ) 6. 8 Q { Recitation Problem Set 9 Ch 8/9 Name: 1. Predict the type of bond (ionic, purely covalent, nonpolar covalent or polar covalent) one would expect to form between the following pairs of elements. Use the table of electronegativity values. Use the arrow notation to indicate which element is the positive end option and which is the negative end of the dipole in the bonds that are polar (length of arrow tells how polar bond is). Indicate the positive and negative ions in ionic compounds. option A. Rb and Cl B. S and S C. C and F D. Ba and S E. N and P F. B and Harrow_forward
- Indicate the polarity of the bond using partial charges (a) N--B (b) N--Oarrow_forwardFor each bond, show the direction of polarity by placing a + sign next to the atom expected to have a partial positive charge and a-sign next to the atom expected to have a partial negative charge. (A) (B) Br (C) For The most polar bond is Submit Answer Try Another Version 4 hem attempts remainingarrow_forwardLogin | Grammarly → C esc 4 Discussion Question 1: Chapter 6 Homework walent and lonic Compound Nomenclature X AW2 Discussion Question 1: Cha x MasteringChemistry: W4 Discu x b Answered: calcium bicarbon: x G pci meaning - Google Search X session.masteringchemistry.com/myct/itemView?assignment ProblemID=190556638&offset=next Q Each ionic compound name will correspond to a single formula. The names of polyatomic ions often need to be memorized. However, ionic compounds with polyatomic ions must still be neutral. Charges are balanced the same way as compounds with single atom cations an anions, such as Li Ammonium chloride 4 Z NH CH NaHCO3 Na₂ NO₂ W S pe into the "Compound not listed" bin. Using the table provided, place the following neutral ionic compounds consisting of polyatomic ions in the bins with their corresponding names. Place compounds that don't belong to any of the compound names displayed on the bin Drag the appropriate items to their appropriate bins. ►View Avallable…arrow_forward
- Benzene (electronic structure, hybridization, bond angles, geometry, complete structureshowing all atoms and bonds, resonance structures, and names of benzene derivatives. clear and short answer pleasearrow_forwardChapter 1 Problem 64 ▼ Part C (CH3)3 C+ Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all hydrogen atoms. Include all lone pairs of electrons. Show the formal charges of all nonhydrogen atoms in the correct structure. [] + Submit C H O Request Answer N S P F Br CI i I X @? More 13arrow_forwardQuestion 5 The Lewis structure for the ion, HCO2, should show bond(s), double bond(s), and lone pair(s). a 2, 1, 5 b 1, 2, 4 c 4, 1, 0 d 3,0, 6 e 2, 2, 4 Please solve correctly. Gpt/ai wrong ans not allowed.arrow_forward
- Which of the following are ionic compounds? [Select all that apply.] K3PO4 O H&PO4 O H2S O MgCl2 NH3 O NH4NO3 CH4arrow_forwardCourse dashboard Which of the following is the correct Lewis structure of HCN? Lütfen birini seçin: A H-c= N: OB. H=C= N: Oc. H-CEN OD. H-CE N: OE H-C-N:arrow_forwardWhich of the following bonds would be classified as nonpolar covalent bonds? [Select all that apply.] O C-C O C-F O F-F O P-H O N-H O C-H O Li-CI O C-Narrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

