
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.47AQAP
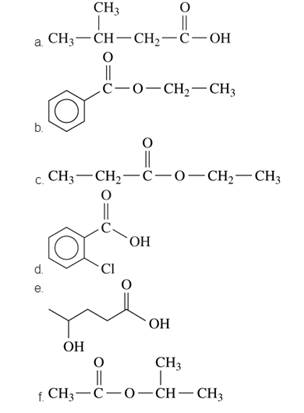
Give the IUPAC and common names, if any, for each of the following compounds: (16.1, 16.4)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
12.42 Which of the following will give a positive Tollens' test? (12.4)
1-propanol
b. 2-propanol
c. hexanal
a.
(12.9) Which of the following has the highest boiling point?
O Both H₂S and H₂O have the same boiling point that is higher than the boiling point of H₂Se.
O H₂Se
O H₂O
O H₂S
12.58 Draw the condensed structural or line-angle formula, if cyclic,
for each of the following: (12.3)
a. formaldehyde
c. 3-methyl-2-hexanone d. 3,5-dimethylhexanal
b. 2-chlorobutanal
Chapter 16 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 16.1 - What carboxylic acid is responsible for the pain...Ch. 16.1 - What carboxylic acid is found in vinegar?Ch. 16.1 - Prob. 16.3QAPCh. 16.1 - Draw the condensed structural formula and give the...Ch. 16.1 - Prob. 16.5QAPCh. 16.1 - Prob. 16.6QAPCh. 16.1 - Draw the condensed structural formula for each of...Ch. 16.1 - Prob. 16.8QAPCh. 16.1 - Prob. 16.9QAPCh. 16.1 - Prob. 16.10QAP
Ch. 16.2 - Prob. 16.11QAPCh. 16.2 - Prob. 16.12QAPCh. 16.2 - Prob. 16.13QAPCh. 16.2 - Prob. 16.14QAPCh. 16.2 - Prob. 16.15QAPCh. 16.2 - Prob. 16.16QAPCh. 16.2 - Prob. 16.17QAPCh. 16.2 - Prob. 16.18QAPCh. 16.2 - Prob. 16.19QAPCh. 16.2 - Prob. 16.20QAPCh. 16.3 - Prob. 16.21QAPCh. 16.3 - Prob. 16.22QAPCh. 16.3 - Prob. 16.23QAPCh. 16.3 - Prob. 16.24QAPCh. 16.3 - Prob. 16.25QAPCh. 16.3 - Prob. 16.26QAPCh. 16.3 - 16.27 Give the IUPAC and common names,if any, of...Ch. 16.3 - Prob. 16.28QAPCh. 16.4 - Prob. 16.29QAPCh. 16.4 - Prob. 16.30QAPCh. 16.4 - Prob. 16.31QAPCh. 16.4 - Prob. 16.32QAPCh. 16.4 - Prob. 16.33QAPCh. 16.4 - Prob. 16.34QAPCh. 16.5 - Prob. 16.35QAPCh. 16.5 - Prob. 16.36QAPCh. 16.5 - Prob. 16.37QAPCh. 16.5 - Prob. 16.38QAPCh. 16.5 - Prob. 16.39QAPCh. 16.5 - Prob. 16.40QAPCh. 16 - Prob. 16.41UTCCh. 16 - Prob. 16.42UTCCh. 16 - Prob. 16.43UTCCh. 16 - Prob. 16.44UTCCh. 16 - Prob. 16.45UTCCh. 16 - Prob. 16.46UTCCh. 16 - Give the IUPAC and common names, if any, for each...Ch. 16 - Prob. 16.48AQAPCh. 16 - Prob. 16.49AQAPCh. 16 - Prob. 16.50AQAPCh. 16 - Prob. 16.51AQAPCh. 16 - Prob. 16.52AQAPCh. 16 - Prob. 16.53AQAPCh. 16 - Prob. 16.54AQAPCh. 16 - Prob. 16.55AQAPCh. 16 - Prob. 16.56AQAPCh. 16 - Prob. 16.57AQAPCh. 16 - Prob. 16.58AQAPCh. 16 - Prob. 16.59AQAPCh. 16 - Prob. 16.60AQAPCh. 16 - Prob. 16.61AQAPCh. 16 - Prob. 16.62AQAPCh. 16 - Prob. 16.63AQAPCh. 16 - Prob. 16.64AQAPCh. 16 - Prob. 16.65CQCh. 16 - Prob. 16.66CQCh. 16 - Prob. 16.67CQCh. 16 - Prob. 16.68CQCh. 16 - Prob. 16.69CQCh. 16 - Prob. 16.70CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Simple alcohols (just containing C's, H's and an OH, where the H on the O is the acidic H) have a pKa similar to water (15.7) hydronium ion (-2)arrow_forwardRank the following substances in order of increasing acidity: Which, if any, of the four compounds is a strong enough acid to react almost completely OH CHyCH3 CHyČCH,ČCH, CH,COH Acetone Pentane-2,4-dione Phenol Acetic acid with NaOH? (pK, 19.3) (pK, = 9) (pK, = 9.9) (pK,4.76) (The pK, of H20 is 15.74.)arrow_forwardplease answer correctly for upvote!!(15.4)arrow_forward
- Give only typing answer with explanation and conclusion Which of the following statements is/are incorrect? (6,25 p) I- In order for an organic compound to be a dyestuff, it must be chromor and auxochrochrome. II- Hydroxyl or amino groups can be given as examples of chromophore groups III- Decrease of chromophore groups in a molecule causes color deepening. A. b) II, III B. a) I only C. d) II only D. c) III onlyarrow_forwardwhat starting materials and reagents could form the indicated product ? Fill in the reaction, acco unt for H indicated as being from the reaction 1.2.3.4.5arrow_forwardCan you help me answer this questions please! (3,4)arrow_forward
- (1)Which is aromatic? (2, 3. 5.arrow_forwardIdentify the main product from the following reaction: I) EtMgBr (1 equlvalent) THF,-78 'C product HO Bu i) H,0* HO НО ОН Et OH EtO Bu Et Bu Et Bu Et Bu HO Bu 2. 3 Select one: 2. 4. 1.arrow_forwardPleaseee answer it correctly the one who answered my questions early is incorrect ithink so. So, please show it and answer it correctly!!!!!!answerr the 2 questions only (7,8)arrow_forward
- ✓ 1 ✓ CaSO4 Screen Reader: Ca S 04 SrCl2 Al2(SO4)3 Screen Reader: A 1 2 left parenthesis S O 4 right parenthesis 3 MgCl2 1. 2. 3. 4. AI(OH)3 + H2SO4 → Screen Reer: A I left parenthesis OH right parenthesis 3 + H2 S 04 Mg(OH)2 + HCI → Screen Reader: Mg left parenthesis O H right parenthesis 2 + HCI → AI(OH)3 + HCI → Screen Reader: A I left parenthesis OH right parenthesis 3 + HCI → NaOH + H3PO4 → Screen Reader: Na O H+H3 P O4arrow_forward(10) (12) H₂0 H+ одосна 3 NaOH H₂0 3arrow_forward(13) 14 15 $ 애 애 1. NaHCO3, H2O 2. H+ Br 애arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY