
Concept explainers
(a)
Interpretation:
The compound
Concept Introduction:
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
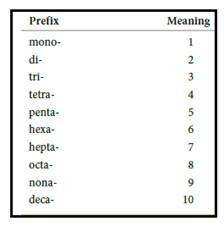
Figure 1
(b)
Interpretation:
The compound
Concept Introduction:
Binary oxygen containing compound are categorised on the basis of oxidation state of their oxide in the compound. A binary compounds has Oxygen with oxidation number of
(c)
Interpretation:
The compound
Concept Introduction:
Naming molecular compounds:
To name binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
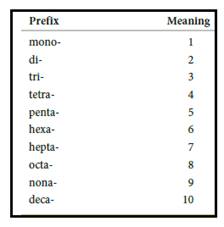
Figure 1
(d)
Interpretation:
The compound
Concept Introduction:
Binary oxygen containing compound are categorised on the basis of oxidation state of their oxide in the compound. A binary compounds has Oxygen with oxidation number of
(e)
Interpretation:
The compound
Concept Introduction:
Binary oxygen containing compound are categorised on the basis of oxidation state of their oxide in the compound. A binary compounds has Oxygen with oxidation number of
(f)
Interpretation:
The compound
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General Chemistry: Atoms First
- 12 8.4Be + 2He → 6C+. 6C+ 0² O 1H 1H 1 On O 0 learrow_forwardWhat is the product of the following reaction? OH 1300 NO2 (a) Ho- (b) (c) (d) no reaction occurs NO2 TON,arrow_forwardPredict which of these compounds are ionic and which are covalent. State your reasoning.(a) BaS(b) CaCO3(c) C6H5Clarrow_forward
- Predict the products from the following chemical reactions. (a) HNO3 + Ba(OH)2 ⟶ (b) CaCl2 + CsOH ⟶ (c) Ammonium Phosphate + Magnesium Sulfate ⟶arrow_forwardGive the name and formula for the acid derived from each of the following anions: (a) perchlorate (b) NO3-(c) bromite (d) H2PO4-arrow_forward3.39 (a) Which of the following molecules can hydrogen bond to another molecule like itself? (b) Which of the following molecules eán hydrogen bond to water? OH СНО NH N. Aarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





