
(a)
Interpretation:
Natural source and a medicinal use of morphine should be stated.
Concept Introduction:
Alkaloids are naturally occurring
E.g. Morphine has analgesic properties, Cocaine has stimulant properties, quinine has antimalarial properties, etc.
Chemical structure of morphine is shown below.
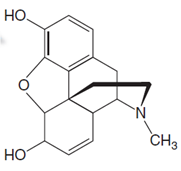
(b)
Interpretation:
Natural source and a medicinal use of quinine should be stated.
Concept Introduction:
Alkaloids are naturally occurring amines derived from plant sources. In general, all alkaloids are bitter in taste. Many alkaloids show pharmacological properties.
E.g. Morphine has analgesic properties, Cocaine has stimulant properties, quinine has antimalarial properties, etc.
Chemical structure of quinine is shown below.
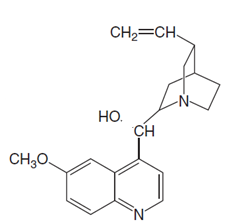
(c)
Interpretation:
Natural source and a medicinal use of atropine should be stated.
Concept Introduction:
Alkaloids are naturally occurring amines derived from plant sources. In general, all alkaloids are bitter in taste. Many alkaloids show pharmacological properties.
E.g. Morphine has analgesic properties, Cocaine has stimulant properties, quinine has antimalarial properties, Atropine has muscle relaxing properties, etc.
Chemical structure of atropine is shown below.
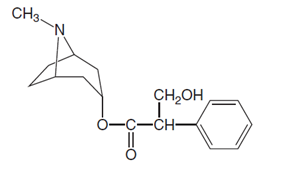
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardDraw a structural formula for each amine. (a) 1,5-Pentanediamine (b) 2-Bromoaniline (c) Tributylaminearrow_forwardExplain the following terms giving one example of each type: (i) Tranquilizers (ii) Synthetic detergents (iii) Food preservativesarrow_forward
- Ester compounds often have a sweet, pleasant odor. Many characteristic fruit scents are largely due to the natural presence of one or more ester compounds. As such, artificial scents for foods are often composed of complex mixtures of various esters. The exact identity and ratio of ingredients that compose a particular scent are closely guarded secrets in the food and fragrance industry. Suppose that you are a chemist working for a company that i creating a new line of air fresheners. The company is considering three scents: apple, pear, and pineapple. The project manager has asked you to prepare the ester compounds that are largely responsible for these scents. The structural formulas for these ester compounds are shown here: Alcohols for Air Freshener Project Molar mass Density Cost, per (g/mL) Reagent (g/mol) 1.00 L methanol 32.04 0.79 $46.20 ethanol 46.07 0.79 $112.00 1-propanol 60.10 0.80 $72.70 1-butanol 74.12 0.81 $72.60 Use the structural formulas of the alcohols and…arrow_forwardSodium borohydride used in the organic lab is a(n) amine carboxylic acid oxidizing agent reducing agentarrow_forwardGive at least 5 examples of biological compounds having an amine functional group and identify the biochemical importance of each compound.arrow_forward
- Give at least 5 examples of biological compounds having a carboxylic acid functional group and identify the biochemical importance of each compound.arrow_forward3. Give the pharmacological property of the following alkaloids. Where do they naturally occur? a. morphine b. quinine c. berberine 4. What are the chemical tests used in identifying alkaloids?arrow_forward2-chloro-1-phenylpropane is reacted with ammonia, producing amphetamine, a widely misused stimulant. Write the chemical equation for the synthesis of amphetamine as described showing the structure of the compounds.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



