
Concept explainers
(a)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are sixteen
Explanation of Solution
The line drawing of the given molecule is:
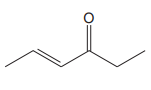
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
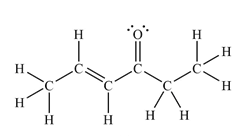
This shows fourteen single bonds, two double bonds, and two lone pairs. Therefore, the molecule contains a total of sixteen
A single bond between two atoms is a
(b)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are sixteen
Explanation of Solution
The line drawing of the given molecule is:
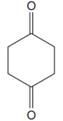
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
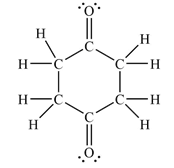
This shows fourteen single bonds, two double bonds, and four lone pairs. Therefore, the molecule contains a total of sixteen
A single bond between two atoms is a
(c)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are twelve
Explanation of Solution
The line drawing of the given molecule is:
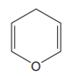
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
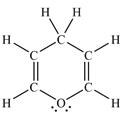
This shows twelve single bonds, two double bonds, and two lone pairs. Therefore, the molecule contains a total of twelve
A single bond between two atoms is a
(d)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are nineteen
Explanation of Solution
The line drawing of the given molecule is:
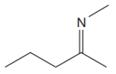
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
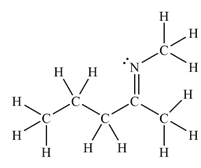
This shows eighteen single bonds, one double bond and one lone pair. Therefore, the molecule contains a total of nineteen
A single bond between two atoms is a
(e)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are eleven
Explanation of Solution
The line drawing of the given molecule is:
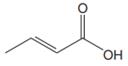
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
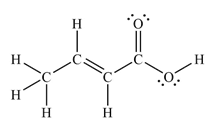
This shows nine single bonds, two double bonds, and four lone pairs. Therefore, the molecule contains a total of eleven
A single bond between two atoms is a
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Below are two sets of resonance structures. Where applicable provide the missing curved arrow notation, lone pair electrons, and nonzero formal charge. Do not delete or add any bonds or atoms.arrow_forwardWould each end of the bond line structure be carbon? Is problem (a) correct?arrow_forwardProblem (1) Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do. CHF=CHF FC CH2 CH;=CH-CH,-CH3 -CHCH, -CHCHCH, CHCH,arrow_forward
- 4)On the following molecules, draw the curved arrows to convert the left-hand resonance structure form to the right-hand resonance structure formarrow_forwardDraw the major resonance structure for the compound shown; include lone pairs of electrons, formal charges, and condensed hydrogen atoms (located in the More menu). Then draw curved arrows to show how this can be converted to the Lewis structure givenarrow_forwardIdentify all functional groups that are present in strychnine, a highly toxic alkaloid used as a pesticide to kill rodents, whose line structure is shown here. What compound class is characteristic of each of those functional groups? Strychninearrow_forward
- Homework problem wants to different structures that have the same molecular formula but different connectivities for c5h12arrow_forwardSee image and include any lone pairs and any charges as wellarrow_forwardProblem What amount (mol) of each ion is in each solution?(a) 5.0 mol of ammonium sulfate dissolved in water(b) 78.5 g of cesium bromide dissolved in water(c) 7.42×1022 formula units of copper(II) nitrate dissolved in water(d) 35 mL of 0.84 M zinc chloridePlan We write an equation that shows 1 mol of compound dissociating into ions. (a) We multiply the number of moles of ions by 5.0. (b) We first convert grams to moles. (c) We first convert formula units to moles. (d) We first convert molarity and volume to moles.arrow_forward
- Draw all reasonable resonance structures for the following compounds. Be sure to show the proper arrows to indicate electron movementarrow_forwardFor each of the following molecules, draw all the valid resonance formsarrow_forwardProblem: (a) Draw six (6) constitutional isomers of C6H1202. (b) Which of the compounds you drew would be the most soluble in water? Explain your choice. (c) Which of the compounds you drew would be the most soluble in hexane? Explain your choice.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
