
Concept explainers
(a)
Interpretation:
The energy corresponding to the IR photons in the given process has to be determined.
Concept introduction:
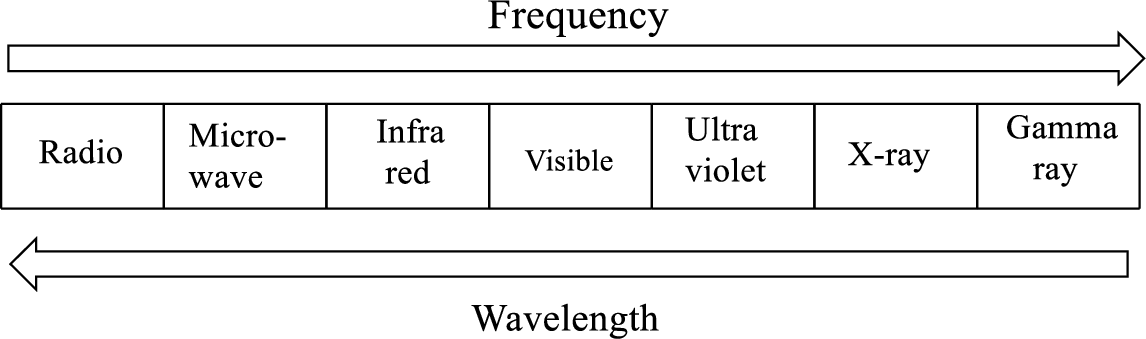
Radiant energy emitted by all matter whose temperature is greater than absolute zero
(
Radiant energy with short wavelength (higher frequency) will have the most energy and as the wavelength get longer, the energy decreases.
The energy of a photon can be calculated using the equation,
(b)
Interpretation:
The correct answer has to be given for the question, what will happens to the energy in the vibrating
Concept introduction:
The energy of a photon can be calculated using the equation,
The energy of a molecule can transferred to any other objects or material through various ways, and these can also spontaneously emitted back to the atmosphere or into space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Chemistry In Context
- hat does temperature measure? Are the molecules in a beaker of warm water moving at the same speed as the molecules in a beaker of cold water? Explain? What is heat? Is heat the same as temperature?arrow_forward16.The frequency of vibration of a bond is a function of which two (2) factors?arrow_forwardBond dissociation energies are often measured by determining the wavelength of ultraviolet photons needed to cause a covalent bond between two atoms to break. A single photon absorbed by a molecule will cause the bond to break between two atoms. If a bond dissociation energy is 450 kJ/mole, what wavelength of photon, in nm, will cause the bond to break?arrow_forward
- What effect does the ozone layer have on the EM waves from the sun? What is currently threatening the ozone layer? Describe the greenhouse effect occurring in Earth's atmosphere? Iarrow_forwardThe greenhouse effect is a natural process that maintains the Earth's temperature within a range that sustains life. Which statement accurately describes the cause of the enhanced greenhouse effect? O a. Burning fossil fuels causes an increase in the amount of atmospheric greenhouse gases, such as CO2, resulting in the enhanced greenhouse effect. O b. Increased amounts of solar energy and infrared radiation are trapped by atmospheric greenhouse gases as a result of the sun burning hotter, which contributes to the enhanced greenhouse effect. O c. Cellular respiration and photosynthesis use CO2 from the atmosphere, reducing the amount of greenhouse gases in the atmosphere. o d. Ultraviolet radiation from cyclic solar flares enhance the greenhouse effect by increasing the amount of thermal radiation absorbed by greenhouse gases in the atmosphere.arrow_forward8) In an exothermic reaction, the enthalpy of the reaction can be calculated by a. Subtracting the sum of all the bond energies of the products from the sum of all the bond energies of the reactants. b. Subtracting the sum of all the bond energies of the reactants from the sum of all the bond energies of the products c. Finding the sum of the enthalpies of formation of all the products and reactants together d. All of the abovearrow_forward
- 1. What is the greenhouse effect? 2. What is good about the greenhouse effect? (Give at least two reasons.) 3. What is the most important greenhouse gas in the troposphere?arrow_forward9.10 The kinetic energy of molecules is often used to induce chemical reactions. The bond energy in an O1 molecule is 8.221019J . Can an O2 molecule traveling at 780 m/s provide enough energy to break the O = O bond? What is the minimum velocity of an O2 molecule that would give a kinetic energy capable of breaking the bond if it is converted with 100% efficiency?arrow_forwardGiven the following data calculate H for the reaction On the basis of the enthalpy change, is this a useful reaction for the synthesis of ammonia?arrow_forward
- Although nitrogen trifluoride (NF3) is a thermally stable compound, nitrogen triiodide (NI3) is known to be a highly explosive material. NI3 can be synthesized according to the equation BN(s) + 3IF(g) BF3(g) + NI3(g) a. What is the enthalpy of formation for NI3(s) given the enthalpy of reaction (307 kJ) and the enthalpies of formation for BN(s) (254 kJ/mol), IF(g) (96 kJ/mol), and BF3(g) (1136 kJ/mol)? b. It is reported that when the synthesis of NI3 is conducted using 4 moles of IF for every 1 mole of BN, one of the by-products isolated is [IF2]+[BF4]. What are the molecular geometries of the species in this by-product? What are the hybridizations of the central atoms in each species in the by-product?arrow_forwardWhat is a driving force? Name two common and important driving forces, and give an example of each. What is entropy? Although the total energy of the universe is constant, is the entropy of the universe constant? What is a spontaneous process?arrow_forward7.86 Nitrogen triiodide, NI3(s) , is unstable and will spontaneously detonate to form a bright purple cloud of nitrogen and iodine gases accompanied with a loud ‘bang,” which suggests a release of energy. Considering the bonds involved, explain why this reaction is energy.arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





