
Concept explainers
(a)
Interpretation: The conjugate base of acetic acid with its two important resonance structuresshould be determined.
Concept introduction: According to Bronsted-Lowry concept, substance that donates proton is termed as acid while that accepts or gains protons is called base. Species formed after loss of protons from acids are known as their respective conjugate bases whereas conjugate acid is produced by addition of protons to base.
(b)
Interpretation: The rank of below bases from highest to lowest potential energy on the basis of their
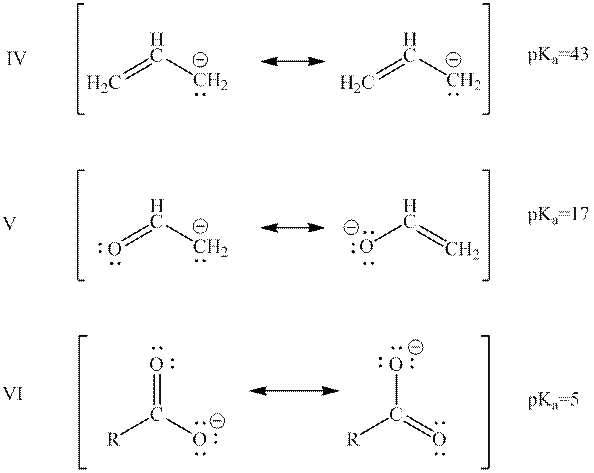
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
(c)
Interpretation: The rank of below anions from highest to lowest potential energy on the basis of resonance should be determined.
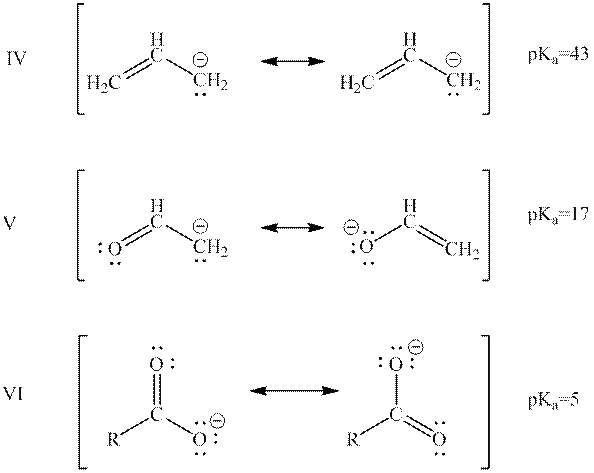
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
(d)
Interpretation: The quantity and quality of resonance structures of below anions can affect their potential energy should be summarized.
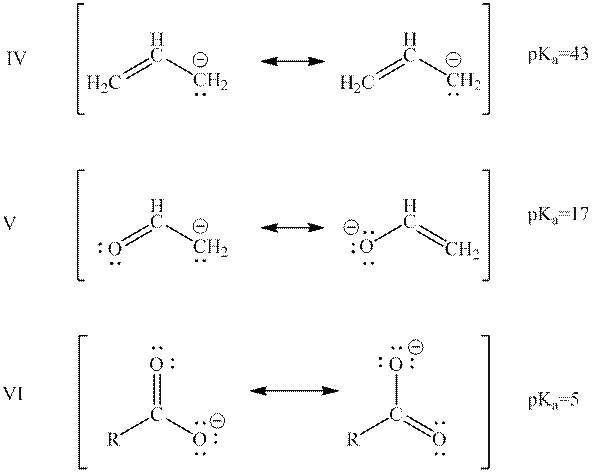
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- 1. An acid-base reaction is shown below. ONa + Base Acid Draw the arrow-pushing mechanism for this reaction. b. Draw the products for the reaction and label them as "conjugate base" and "conjugate I | acid." Be sure to assign formal charges. c. Consider the base and conjugate base. Using your four tools, identify the weakest conjugate base and put a box around it. Which tool did you use to determine your answer? d. Using the pKa chart in you lecture notes, label the acid and conjugate acid with their pK, values. Which functional group is the stronger acid? e. Equilibrium moves away from the stronger acid (it wants to dissociate) and toward the more stable conjugate base (lower energy side). Based on your answers to the previous questions, which side of the reaction is favored by equilibrium, left or right? HIN:arrow_forward6. Classify each acid base reaction as a Brønsted or Lewis pair. Label the acid (or electrophile) and the base (or nucleophile). Draw a curved arrow to show how electrons moved to form the new bond(s). i CH3Li -H + Na + CH3Li NaNH, OH Br H3C- O Lit O Lit 핸 Na CH4 NH3 + NaBrarrow_forwardDraw the structure of the conjugate base of water. (Note that it does not appear in Figure 4.11).arrow_forward
- For each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forwardDraw the products of each acid-base reaction. EXAMPLE: a. b. CH3CH₂ propanoic acid OH || + CH3CH₂CH₂ OH + K+ OH This proton is transferred from the acid to the base. NaOH + base Na₂CO3 + H-O-H CH₂CH₂ O K+ The carboxylate anion is formed as a potassium salt.arrow_forwardAde ed arrows to show the Arte attack the proton of the acid (breaking the X-H bond * Identify the nucleophile and electrophile ectants but the charge will be on a different molecule.) Circle the conjugate base. charges of any lons OK a) HO, b) кон SH c) NO. ONa HO + Approximate pK, values of Important organic functional groups pk, Protonated Cacborylic alcohol R. -2 5. 10 Phenol 16 Alcohol 25 Alkyne 38 Amine acid 44 Alkene 50 1. H OH R-OH R -H Alkane R. R. он R H ROH Protonated N amine H. H Thiol R-SHarrow_forward
- Add curved arrows to show the forming and breaking of bonds in the reaction below. H H H Add/Remove step C H H Harrow_forwardDraw the curved arrows showing a proton transfer reaction, and draw the products of that proton transfer. Do not include the Li+ counterion, and lone pairs are not required in the products. Do not use abbreviations such as Me or Ph. (x) O. 0 H. %3D P. OH + H,C CI H;C Br I +t +# +Larrow_forwardSelect the outcome of the following reaction. Br2 Br Br only Br only Brm Br Brarrow_forward
- Part IV: Draw the mechanism and the energy diagram for the reaction shown below. Include any resonance structures for the intermediates of the reaction. Br A OHarrow_forward3. Use curved arrows to show electron movement in the reactant side and draw the product/s of the Lewis acid-base (nucleophile-electrophile) reaction. Label the nucleophile and the electrophile. Draw in all lone pairs and charges where appropriate. -co 요arrow_forwardWhich reaction corresponds to the Kb for HSO4− ? a. H2SO4 ⇌ H+ + HSO4− b. SO42− + H2O ⇌ OH− + HSO4− c. HSO4− + H2O ⇌ OH− + H2SO4 d. HSO4− ⇌ H+ + SO42− e. HSO4− + OH− ⇌ H2O + SO42−arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
