
Inorganic Chemistry
5th Edition
ISBN: 9780321811059
Author: Gary L. Miessler, Paul J. Fischer, Donald A. Tarr
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 5.4, Problem 5.5E
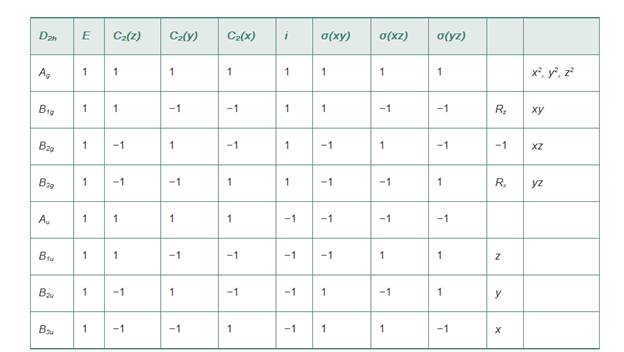
Using the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider the molecular orbital diagram below for carbon dioxide. Refer to the molecular orbitals using the letters on the far right of the diagram
(a) Identify the HOMO to LUMO transition by reporting the initial and final orbitals for each transition. Then calculate the wavelength [in nanometers] and frequency [in Hz] for the photon involved in
the transition if the energy change is 2.0 eV.
(b) Identify the m\pitT-to-TT\piT* and n-to-TT\piTT* transitions by reporting the initial and final orbitals for each transition. Your textbook states than typical transition energies are 7eV (TT\piTT-to-TT\piTT*) and
4 ev (n-to-TT\pir*). Even though the diagram here does not give numerical values for energies, you can still make use of this typical behavior to give you confidence in your answers.
DFT BILYP 6-311++G(d.p)
G
2Px 2Py 2P
2P 2Py 2P
11
E
25
D
В
A
one pai
20
Please arrange the following: Kr, Cl2, H2, N2, Ne, and O2 in order of increasing polarizability and explain your reasoning
Relate the energies of the orbitals found (these we will use for building Molecular Orbital energy level diagrams) to Z*, also known as Zeff, determined by Slater’s rules Please show calculations for the following:
H (1s electron)
He (1s electron)
Chapter 5 Solutions
Inorganic Chemistry
Ch. 5.1 - Repeat the process in the preceding example for...Ch. 5.2 - Prob. 5.2ECh. 5.3 - Use a similar approach to the discussion of HF to...Ch. 5.4 - Sketch the energy levels and the molecular...Ch. 5.4 - Using the D2h character table shown, verify that...Ch. 5.4 - Using orbital potential energies, show that group...Ch. 5.4 - Prob. 5.7ECh. 5.4 - Prob. 5.8ECh. 5.4 - Prob. 5.9ECh. 5.4 - Use the projection operator method to derive...
Ch. 5.4 - Determine the types of hybrid orbitals that are...Ch. 5.4 - Determine the reducible representation for all the...Ch. 5 - Expand the list of orbitais considered in Figures...Ch. 5 - On the basis of molecular orbitals, predict the...Ch. 5 - On the basis of molecular orbitals, predict the...Ch. 5 - Compare the bonding in O22,O2 and O2 Include Lewis...Ch. 5 - Although the peroxide ion, O22 and the acetylide...Ch. 5 - High-resolution photoelectron spectroscopy has...Ch. 5 - a. Prepare a molecular orbital energy-level...Ch. 5 - a. Prepare a molecular orbital energy-level...Ch. 5 - NF is a known molecule a. Construct a molecular...Ch. 5 - The hypofluorite ion, OF can be observed only with...Ch. 5 - Prob. 5.11PCh. 5 - Although KrF+ and XeF+ have been studied, KrBr+...Ch. 5 - Prepare a molecular orbital energy level diagram...Ch. 5 - Methylene, CH2 plays an important role in many...Ch. 5 - Beryllium hydride, BeH2 is linear in the gas...Ch. 5 - In the gas phase, BeF2 forms linear monomeric...Ch. 5 - For the compound XeF2 do the following: a. Sketch...Ch. 5 - TaH5 has been predicted to have C4v symmetry, with...Ch. 5 - Describe the bonding in ozone, o3 on the basis of...Ch. 5 - Describe the bonding in SO3 by using group theory...Ch. 5 - The ion H3+ has been observed, but its structure...Ch. 5 - Use molecular orbital arguments to explain the...Ch. 5 - Prob. 5.23PCh. 5 - Prob. 5.24PCh. 5 - The isomenc ions NSO (thiazate) and SNO...Ch. 5 - Apply the projection operator method to derive the...Ch. 5 - Apply the projection operator method to derive the...Ch. 5 - A set of four group orbitals derived from four 3s...Ch. 5 - The projection operator method has applications...Ch. 5 - Although the cl2+ ion has not been isolated, it...Ch. 5 - BF3 is often described as a molecule in which...Ch. 5 - SF4 has C2v symmetry. Predict the possible...Ch. 5 - Consider a square pyramidal AB5 molecule. Using...Ch. 5 - Prob. 5.34PCh. 5 - For the molecule PCl5 : a. Using the character...Ch. 5 - Molecular modeling software is typically capable...Ch. 5 - Prob. 5.39PCh. 5 - Calculate and display the orbitals for the linear...Ch. 5 - Prob. 5.41PCh. 5 - Prob. 5.42PCh. 5 - Prob. 5.43PCh. 5 - Diborane, B2H6 , has the structure shown. a. Using...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The B2 molecule is paramagnetic; show how this indicates that the energy ordering of the orbitals in this molecule is given by Figure 6.18a rather than 6.18b.arrow_forwardThe molecule PtCl 2 - is an important square planar starting material in synthetic inorganic chemistry. Using the p orbitals of Cl as the basis find the irreducible representations of the p orbitals and indicate which orbitals could possibly overlap with any of the orbitals on Pt . Assume that the pz orbitals on Cl are orientated in the same direction as the principle rotation axis, px orbitals are pointed at the Pt , and the py orbitals are orthogonal to the px orbitals. Show the characters of each orbital and identify which orbitals on Pt would match from a pure symmetry standpoint.arrow_forwardWhat is the reducible representation for BH4+arrow_forward
- Which of the d orbitals can form pi / pi*? and Which of the d orbitals can form delta / delta * ? Thank you.arrow_forwardHuckel theory: All atomic orbital coefficients in the lowest energy MO (Ψ1) should have the same value – explain why?arrow_forwardWhat is the order with respect to A2 and B2?arrow_forward
- 2)How many of the following elements have paramagnetic adamant in the ground eletronic state? We do not need to consider irregular electronica configurations.  Ca Zr Zn In Krarrow_forwardgive application of group theoryarrow_forward(a) Given the operator = -0 C₁¹ show that [‚Â] = 0 =0 (b) Decompose the representation below into the irreducible representations of C3v. C3v T E 17/0 2C3 1 30₂ 1 ] ] (c) With the use of diagrams, find the representation of the C₂ point group to which a day orbital belongs. ]arrow_forward
- Does SP have p orbitals?arrow_forwardThe group C2h consists of the elements E, C2, σh, i. Construct the group multiplication table. Give an example of a molecule that belongs to the group.arrow_forwardDraw the overlapping orbitals of (a) 2 p orbitals then give 2 examples of compounds that exhibits these kinds of orbital overlapping. ; (b) 1 s and 1 p orbitals; (c) two s orbitalsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER
Gerade and Ungerade Molecular Orbitals. (SYMMETRY OF MOLECULAR ORBITALS); Author: Edmerls;https://www.youtube.com/watch?v=dPY-lT5LN60;License: Standard YouTube License, CC-BY
Symmetry and chemical bonding part – 5 Molecular orbital formation (CHE); Author: Vidya-mitra;https://www.youtube.com/watch?v=g-42GmpBu0I;License: Standard Youtube License