
Concept explainers
a)
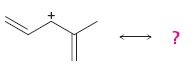
Interpretation:
All the resonance forms that would stabilize the carbocation given are to be drawn.
Concept introduction:
Different resonance forms that stabilize a molecule differ only in the placement of their bonding or nonbonding electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another.
To draw:
All the resonance forms that would stabilize the carbocation given.
b)
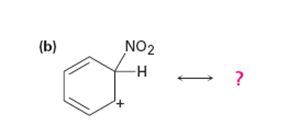
Interpretation:
All the resonance forms that would stabilize the carbocation given are to be drawn.
Concept introduction:
Different resonance forms that stabilize a molecule differ only in the placement of their bonding or nonbonding electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another.
To draw:
All the resonance forms that would stabilize the carbocation given.
c)
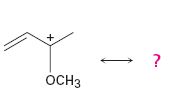
Interpretation:
All the resonance forms that would stabilize the carbocation given are to be drawn.
Concept introduction:
Different resonance forms that stabilize a molecule differ only in the placement of their bonding or nonbonding electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another.
To draw:
All the resonance forms that would stabilize the carbocation given.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- • Question 6: Which alkene in each pair is more stable? a. b. CH3CH₂ H C=C or CH₂CH3 H or CH3CH₂ H H CH₂CH3 2 C. CH3 or CH3arrow_forwarda) This alkene can be prepared via Wittig reaction. Draw structural formulas for the aldehyde and the Wittig reagent. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Draw the Wittig reagent in the ylide form. • If more than one combination of Wittig reagent and aldehyde is possible, draw only one set. b) How would you convert the alkene to a epoxide? MCPBA c) How many chiral centers are present in the epoxide? V How many stereoisomers are possible in the epoxide that you formed in b)?arrow_forwardHow many moles of Bra are required to completely halogenate the alkene?A. One moleB. Two molesC. Three molesD. Four moles What is the expected arrangement of the bromine atoms relative to each other amongthe carbon involved in pi bonding?A. anti-conformationB. syn-conformationC. trans-configurationD. cis-configuration What happens to bromine when it is adjacent to an alkene during a chemical reaction?A. Bromine becomes stable. (? kasi before brown siya/acidic tas naging colorless? Jk ewan)B. Bromine becomes polarized.C. Bromine becomes hybridized.D. Bromine becomes acidic. The relative arrangement of bromine atoms in the product is primarily due to:A. ElectronegativityB. RepulsionC. Hydrogen bondingD. Atomic weightWhat is your observation after the reaction?A. A yellow flame is produced.B. Bromine water decolorizes.C. The alkene becomes denser.D. A brown precipitate forms.arrow_forward
- Zaitsev's rule is useful in selecting which carbon adjacent to a carbocation will form the double bond in the alkene product. True or Falsearrow_forwardWhich of the following concepts explains why a tertiary carbocation is more stable than a primary carbocation? a. Hyperconjugation b. Resonance c. Electronegativity T d. he octet rulearrow_forwardDraw a stepwise mechanism that shows how all three alcohols are formed from the bicyclic alkene. $*$·$·$ OH H₂O H₂SO4 OH OHarrow_forward
- Structures with carbocations can often also have resonance forms. Draw the other resonance structure for the moleculearrow_forward5. For each of the following pairs, circle the most stable alkene and provide the name for each of them. X VS. 2.0 VS. VS.arrow_forward6. Use arrow pushing to draw the product of the resulting reaction. If no reaction occurs, then write in NR. OH Na₂Cr₂O7 H₂SO4, H₂Oarrow_forward
- Determining the Relative Energy of Resonance Structures and the Hybrid Draw a second resonance structure for carbocation A, as well as the hybrid of both resonance structures. Then use Rules [1]–[3] to rank the relative stability of both resonance structures and the hybrid.arrow_forwardCHCHS CH, CH CH CHCH, A D Which is the most stable alkene? Choose. Which is the least stable alkene? Choose.arrow_forwardDraw the skeletal structure of each product for each reaction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
