
Concept explainers
Answer the following questions about each of the hydroxy
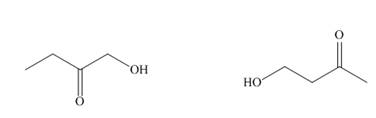
A B
a. What is the molecular ion in the mass spectrum?
b. What IR absorptions are present in the
c. How many lines are observed in the
d. How many signals are observed in the
e. Give the splitting observed for each type of proton as well as its approximate chemical shift.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Living By Chemistry: First Edition Textbook
CHEMISTRY-TEXT
Organic Chemistry - Standalone book
- Draw all the constitutional isomers of octane. Name each one. For each isomer: a. State the number of different sets of carbon in the structure b. State whether each set of equivalent carbons would produce an up or down signal in the DEPT-135 NMR experiment. c. State whether each set of equivalent carbons would produce a singlet, doublet, triplet, or quartet in the Off-Resonance Decoupled Experimentarrow_forward3. Nuclear Magnetic Resonance Spectroscopy a. Draw the 'H NMR spectrum for the following compound. CIarrow_forwardA B. C. H. CH Br HC-C-CH CHy CH3-C-CH H-C-CH,CH, CH CH, Which of the following compounds give four major peaks in their 1H NMR spectra? OA and B. Band C. A and C. O All of themarrow_forward
- Match the spectra to the structures: 3-bromotoluene and methyl 4-methoxybenzoate. CH 3 3-bromotoluene 200 Spectrum B 200 Br (ignore the CDCI3 signal) CH3O methyl 4-methoxybenzoate OCH3 Spectrum A 100 100arrow_forwardWhich of these molecules is the best IR spectrum A. 3- Phenypropanoic acid B. 2-Nethy1-2 - penatanol C none of these D. Propiophenonearrow_forward2. Draw the 1HNMR spectrum of 1-Methoxyhexane. Then answer the following questions in reference to your1H NMR spectrum of 1-Methoxyhexane. a. How many types of H(signals)? b. What types of H(chemical shift)? c. How many H of each type are there (integration)? d. What are the connectivity (coupling patterns)?arrow_forward
- Answer the following questions about each of the hydroxy ketones: 1-hydroxybutan-2-one (A) and 4-hydroxybutan-2-one (B). a.) How many signals are observed in the 1H NMR spectrum?b.) Give the splitting observed for each type of proton as well as its approximate chemical shift.arrow_forwardA. Predict the splitting pattern of the compound (a) in the 1H1H NMR spectrum. B. Predict the splitting pattern of the compound (b) in the 1H1H NMR spectrum. C. Predict the splitting pattern of the compound (c) in the 1H1H NMR spectrum. D. Predict the splitting pattern of the compound (d) in the 1H1H NMR spectrum.arrow_forwardWhat are the major IR absorptions in the functional group region for each compound? a. d. b. O- CH3O C. е. Но capsaicin (spicy component of hot peppers)arrow_forward
- Which pair of molecules would be the most difficult to distinguish using IR spectroscopy? Select one: a. benzoic acid and phenol b. 1-pentanol and 1-pentene C. aniline and phenol d. pentane and hexane e. 2-butanone and butanalarrow_forward6. IR spectroscopy experiment is used to evaluate the success of the reaction. The IR spectra for acetaminophen and phenacetin are shown below. a. Identify which spectrum is for acetaminophen and which is for phenacetin. b. Label the key bond/functional group signals on each spectrum. (eg N-H, C=0, O-H, C=C, C(sp2)-H, C(sp3)-H) OH N H acetaminophen phenacetin 100.0 90 80 70 60 30 40 30 20 10 0.0 40000 3000 2000 1500 1000 400.0 em-1 LOD D 4D00 3000 s0o 2000 1500 1000arrow_forwardEach of the following compounds exhibit a single 1H NMR peak. Approximately wherewould you expect each compound to absorb (give your answer in a range of chemical shift)?a. Cyclohexaneb. Acetonec. Benzened. Dichloromethanee. Trimethylaminearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





