
Concept explainers
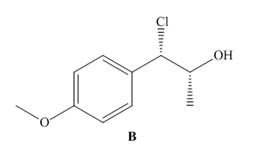
Reaction of unknown A with
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
Chemistry: The Molecular Nature of Matter
Essential Organic Chemistry (3rd Edition)
Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- Reaction between this aldehyde and ketone in base gives a compound A with the proton NMR spectrum: ô 1.10 (9H, s), 1.17 (9H, s), 6.4 (1H, d, J 15), and 7.0 (1H, d, J 15). What is its structure? (Don't forget stereochemistry!). When this compound reacts with HBr it gives compound B with this NMR spectrum: õu 1.08 (9H, s), 1.13 (9H, s), 2.71 (1H, dd, J 1.9, 17.7), 3.25 (dd, J10.0, 17.7), and 4.38 (1H, dd, J 1.9, 10.0). Suggest a structure, assign the spectrum, and give a mechanism for the formation of B. H base C11H₂00 HBr B C11H₂1 Broarrow_forwardA compound reacts with methylmagnesium bromide followed by acidification to form the product with the following 1H NMR spectrum. Identify thecompound.arrow_forwardReaction of p-cresol with two equivalents of 2-methylprop-1-ene affords BHT, a preservative with molecular formula C15H24O. BHT gives the following 1H NMR spectral data: 1.4 (singlet, 18 H), 2.27 (singlet, 3 H), 5.0 (singlet, 1 H), and 7.0 (singlet, 2 H) ppm. What is the structure of BHT? Draw a stepwise mechanism illustrating how it is formed.arrow_forward
- Compound A shows a molecular peak of 56 in Mass Spectrum. When compound A is treated with HBr and peroxides, it produces compound B, which shows twin molecular peaks with weights 136 and 138 in 1:1 ratio. The 'H NMR spectra of both compounds shown below. Identify the structures of A and B in the respective boxes. 2H Compound A 'H NMR of Compound A (integration is listed next to peaks) لد HBr peroxides 2H PPM 'H NMR of Compound B (integration is listed next to peaks, expansions in above) М 1H Compound B PPM 6H 6Harrow_forward(b) Reaction between an aldehyde and ketone (see below), in base gives a compound A with spectrum: 8 1.10 (9H, s), 1.17 (9H, s), 6,4 (1H. d. J 15 Hz), 7.0 (1H, d. J 15 Hz). What is its structure? When this compound reacts with HBr it gives compound B with this NMR spectrum: õ 1.08 (9H, s), 1.13 (9H, s), 2.71 (1H, dd, J 1.9 Hz, 17.7 Hz), 3.25 (1H, dd, J 10.0 Hz, 17.7 Hz), 4.38 (1H, dd, J 1.9 Hz, 10.0 Hz). Suggest a structure, assign the spectrum and give a mechanism for the formation of compound B. H. base HBr B C1H200 CH21Broarrow_forward8 8. Correlate each signal in the following 'H NMR for ethyl cinnamate with a particular set of hydrogens. Be sure to explain the location of the signal, the integration pattern, and the splitting pattern for all peaks. 'H NMR (300 MHz, DMSO): 1.21 (3H, t), 4.06 (2H, q), 6.31 (1H, d), 7.48 (1H, d), 7.33 (1H, t), 7.38 (2H, m), 7.54 (2H, d). 7 6 5 4 PPM 3 2 1 X 0arrow_forward
- Identify products A and B from the given 1H NMR data. Treatment of CH2=CHCOCH3 with one equivalent of HCl forms compound A. A exhibits the following absorptions in its 1H NMR spectrum: 2.2 (singlet, 3H), 3.05 (triplet, 2 H), and 3.6 (triplet, 2 H) ppm. What is the structure of A?arrow_forwardDetermine the structure of the compound with the following spectral data. You must show your reasoning for full credit. (8pts) You must show your reasoning. MS: M+ (164) and M++2 in 1:1 ratio 1H-NMR: 3.5 (2H, triplet) 1.65 (2H, triplet) 0.9 (9H, singlet)arrow_forwardPropose a structure consistent with each set of data Compound A: Molecular formula: C8H10OIR absorption at 3150−2850 cm−11H NMR data: 1.4 (triplet, 3 H), 3.95 (quartet, 2 H), and 6.8–7.3(multiplet, 5 H) ppmarrow_forward
- An unknown compound D exhibits a strong absorption in its IR spectrumat 1692 cm−1. The mass spectrum of D shows a molecular ion at m/z =150 and a base peak at 121. The 1H NMR spectrum of D is shown below.What is the structure of D?arrow_forwardCompound X (molecular formula C10H120) was treated with NH2NH2, ¯OH to yield compound Y (molecular formula C10H14). Match the 1H NMR spectra of X and Y to the corresponding structures of X and Y. Compound NH2NH2 Compound 'H NMR of X 6 H OH Y 1 H 5H 8. 6. 4 ppm or H NMR of Y 6 H 2H 5H 1 H multiplet multiplet 8. 6. 4. 3. 1 nnm 2. 2. 3, O:arrow_forwardThe only organic compound obtained when compound Z undergoes the following sequence of reactions gives the 1H NMR spectrum shown. Identify compound Z.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





