
Concept explainers
Cyclohex-
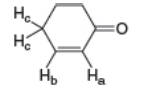
cyclohex-
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Organic Chemistry - Standalone book
Chemistry & Chemical Reactivity
Chemistry: Matter and Change
Organic Chemistry (9th Edition)
Chemistry In Context
Chemistry: The Central Science (14th Edition)
- Use Apendix C or Figure 18.9 to determine whether Kc > 1 or Kc < 1 for each reaction. (a) H2SO3 + NH3 HSO3- + NH4+ Kc < 1 or Kc > 1 (b) HCN + HCO3- H2CO3 + CN - Kc < 1 or Kc > 1arrow_forwardDraw an isomer of difluorocyclopropane that has (a) one proton environment; (b) two proton environments; (c) three proton environments.arrow_forward(a) Rank each proton shown below in terms of relative chemical shift in the ¹H NMR spectrum. (1 = most upfield, 3 = most downfield) (i) (iii) H JO [ Ⓒarrow_forward
- In Section 15.5c, we learned that the frequency of a C=0 stretch decreases when the c=0 bond is conjugated to a C=C bond. Draw the pertinent resonance contributor of a conjugated carbonyl (C=C-c=0) and, based on the resulting resonance hybrid, explain why the frequency decreases.arrow_forwardConsider the following three compounds: CH,=CH, CH,=CH-CH=CH, CH,=CH-CH=CH-CH=CH, (1) (2) (3) Compound 1 contains a simple isolated carbon-carbon double bond, but the other two have conjugated double bonds. (i) Which compound will have the largest absorption (max) Value? (ii) Give a reason for your answer in part (b)(i).arrow_forwardWhich of the underlined protons in each pair absorbs farther downfield: (a) CH,CH,CH, or CH;OCH3; (b) CH;OCH3 or CH3SCH3?arrow_forward
- 1 (a) In the following reactions, (1) LIAIH, A (2) H2O mCPBA B (i) Draw the structure of compounds A and B. (ii) For each reaction, explain the type of reaction involved. (iii) Explain the successful transformation of compound A using mass spectra.arrow_forwardA compound of formula C20H29NO absorbs 4 molar equivalents of hydrogen. How many rings does it contain?arrow_forward3. For the given molecules 1 H NMR (a) label all the hydrogen atoms, (b) State how many signals are possible (c) Predict chemical shift values (d) and Multiplicity. CH, *CH, 1-pentene 3-methylbutanal p-ethylanisole HO, Br butanoic acid 2-bromobutane diethyl malonate OH Br 1-propanol 1-bromopropane diethyl ethylmalonatearrow_forward
- The chemical shifts of the a protons on cyclohexanone and cyclobutanone are as shown here: (a) Which a carbon has a greater effective electronegativity? (b) Can you explain why, using arguments of s character and p 2.4 ppm 3.0 ppm character? Cyclohexanone Cyclobutanonearrow_forwardThe1H NMR spectrum of CH3OH recorded on a 500 MHz NMR spectrometer consists of two signals, one due to the CH3 protons at 1715 Hz and one due to the OH proton at 1830 Hz, both measured downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) Do the CH3 protons absorb upfield or downfield from the OH proton?arrow_forward2. Hexamethylbenzene undergoes free-radical chlorination to give one monochlorinated product (C₁2H17C1) and four dichlorinated products (C12H16C12). These products are easily separated by GC-MS, but the dechlorinated products are difficult to distinguish by their mass spectra. Draw the monochlorinated product and the four dichlorinated products, and explain how ¹3C NMR would easily distinguish among these compounds. (6 pts) Cl₂/hv Ch/v monochlorinated product dichlorinated products CIarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





