
Concept explainers
(a)
Interpretation:
Preparation of 2-bromo-2-methyl from 2-methylpropane has to be shown.
Concept Introduction:
Radical or free radical: The unpaired valence electron of an atom, molecule, or ion is called as radical.
In a halogenation reaction, one or more halogen atoms are introduced into an organic compound. Generally, these reactions are initiated in the presence of light or heat.
Bromination:
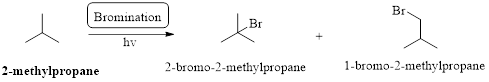
(b)
Interpretation:
Preparation of 2-methyl-1- propene from 2-methylpropane has to be shown.
Concept Introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
In a halogenation reaction, one or more halogen atoms are introduced into an organic compound. Generally, these reactions are initiated in the presence of light or heat.
Bromination:
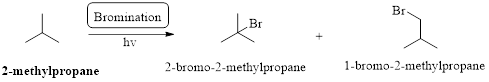
(c)
Interpretation:
Preparation of 2-Iodo-2-methylpropane form 2-methylpropane has to be shown.
Concept Introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
In a halogenation reaction, one or more halogen atoms are introduced into an organic compound. Generally, these reactions are initiated in the presence of light or heat.
Bromination:
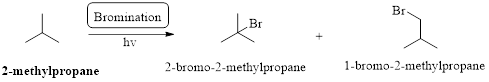
Markovnikov’s rule:
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Essential Organic Chemistry (3rd Edition)
- Consider the structure of cyclohexene, if it undergoes epoxidation followed by exposure to water, which of the following final product is formed? a. Cyclohexan-1,2,-diol b. Cyclohexane c. Cyclohexanone d. Hexan-1,-6-dioic acidarrow_forwardwrite the reaction in which chlorine-2 butane is formed as a result of the action of one of the following compounds a) butene-1 with chlorine b) butene-1 with HCl c) butene-2 with chlorine d) butadiene-1,3 with HClarrow_forward9. What is the major organic product obtained from the following reaction? a. 1 b. 2 c. 3 d. 4 OH H₂SO4 3 SH 2arrow_forward
- What are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. benzoic acid and ethanol b. phenylic acid and ethanol c. ethanoic acid and benzene d. acetic acid and toluene e. phenylic acid and methanolarrow_forwarddraw and write the chemical equation of the following situations: 1. Combustion of acetylene 2. Reaction of acetylene with Potassium Permanganate 3. Reaction of acetylene with cuprous chloride in ammoniaarrow_forwardHow can the following compounds be prepared from 3,3-dimethyl-1-butene? a. 2,3-dimethyl-2-butanol b. 3,3-dimethyl-2-butanolarrow_forward
- Esters are formed by the reaction of carboxylic acids and acid in the presence of A. Alkanes B. Alcohols C. Alkenes D. Aldehydes E. Alkynesarrow_forwardDefine the Properties of Alkyl Halides ?arrow_forwarddraw and write the chemical equation of the following situations: 1. Preparation of acetylene 2. Reaction of acetylene with lime waterarrow_forward
- A process by which a hydrocarbon replaces one of the Hydrogen atom in the benzene ring. A. Acylation B. Alkylation C. Halogenation D. Sulfonationarrow_forward2) Please complete the following syntheses A) Propene to 2-methyl-pent-2-ene B) pentan-2-amine to 3-bromopentanearrow_forwardWhich substances can be obtained by cracking hydrocarbons? A ethanol and ethene B ethanol and hydrogen C ethene and hydrogen D ethene and poly(ethene)arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


