
Concept explainers
(a)
Interpretation:
The structural formula of keto form for the given enol form should be drawn.
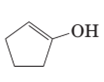
Concept Introduction:
A carbonyl compound that has a-hydrogen on a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
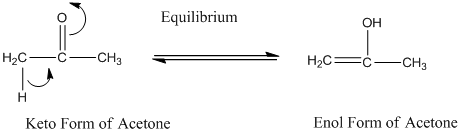
a- hydrogen is the hydrogen which attached to a-carbon for a carbonyl compound. a-carbon is carbon adjacent to the carbonyl carbon. If a compound don’t have any a hydrogen is does not undergoes Keto-Enol tautomerism.
(b)
Interpretation:
The structural formula of keto form for the given enol form should be drawn.
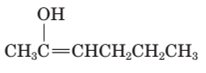
Concept Introduction:
A carbonyl compound that has a-hydrogen on a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
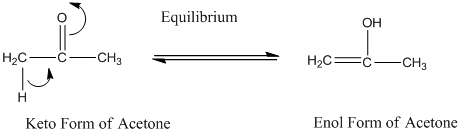
a- hydrogen is the hydrogen which attached to a-carbon for a carbonyl compound. a-carbon is carbon adjacent to the carbonyl carbon. If a compound don’t have any a hydrogen is does not undergoes Keto-Enol tautomerism.
(c)
Interpretation:
The structural formula of keto form for the given enol form should be drawn.
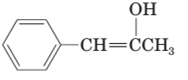
Concept Introduction:
A carbonyl compound that has a-hydrogen on a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
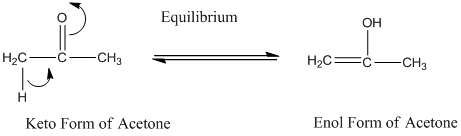
a- hydrogen is the hydrogen which attached to a a-carbon for a carbonyl compound. a-carbon is carbon adjacent to the carbonyl carbon. If a compound don’t have any a hydrogen is does not undergoes Keto-Enol tautomerism.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-47 What is the characteristic structural feature of a hemiacetal? Of an acetal?arrow_forwardGive the IUPAC name for each ketone.arrow_forward17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward
- 17-29 Why can’t two molecules of acetone form a hydrogen bond with each other?arrow_forward17-79 Write an equation for each conversion. (a) 1-Pentanol to pentanal (b) 1-Pentanol to pentanoic acid (c) 2-Pentanol to 2-pentanone (d) 2-Propanol to acetone (e) Cyclohexanol to cyclohexanonearrow_forward16-28 Following is the structural formula of metformin, the hydrochloride salt of which is marketed as the antidiabetic medication Glucophage. Metformin was introduced into clinical practice in the United States in 1995 for the treatment of type 2 diabetes. More than 25 million prescriptions for this drug were written in 2000, making it the most commonly prescribed brand-name diabetes medication in the nation. NH NH H3(\ 3 N N Nh2ch3 h Metformin Complete the Lewis structure for metformin, showing all valence electrons. Which nitrogen is the most likely site of protonation? Draw the structural formula of Glucophage.arrow_forward
- 18-18 Propanoic acid and methyl acetate are constitutional isomers, and both are liquids at room temperature. One of these compounds has a boiling point of 141°C; the other has a boiling point of 57°C. Which compound has which boiling point? Explain.arrow_forwardWhy is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forwardHow to name an acyclic ketone using IUPAC rules ?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





