
(a)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an
Mechanism for NaBH4 reduction explained below.
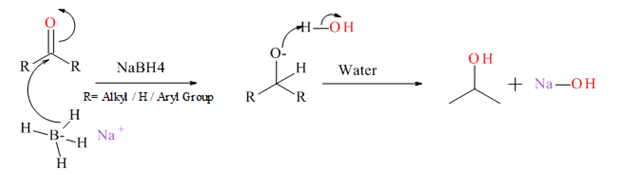
(b)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an aldehyde or ketone reduced to give the corresponding 1° or 2° alcohol. There is several type of reducing agents such as Pd /H2, NaBH4, LiAlH4 etc.
Mechanism for NaBH4 reduction explained below.
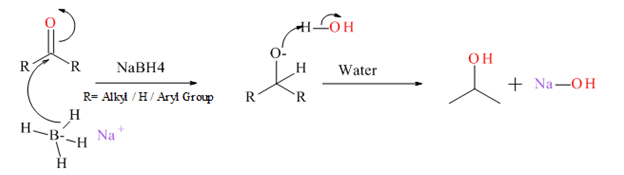
(c)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an aldehyde or ketone reduced to give the corresponding 1° or 2° alcohol. There are several type of reducing agents such as Pd /H2, NaBH4, LiAlH4 etc.
Mechanism for NaBH4 reduction explained below.
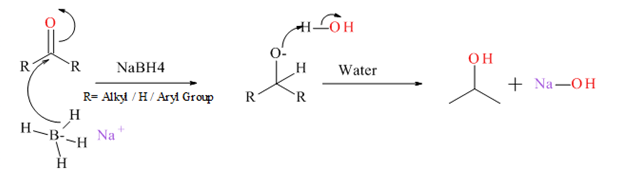
(d)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an aldehyde or ketone reduced to give the corresponding 1° or 2° alcohol. There are several type of reducing agents such as Pd /H2, NaBH4, LiAlH4 etc.
Mechanism for NaBH4 reduction explained below.
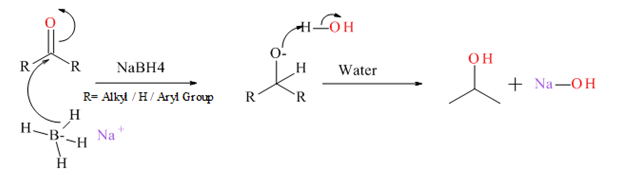
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward14-31 Compare the acidity of alcohols and phenols, which are both classes of organic compounds that contain an —OH group.arrow_forward17-18 Draw structural formulas for these ketones. (a) Ethyl isopropyl ketone (b) 2-Chiorocyclohexanone (c) 2,4-Dimethyl-3-pentanone (d) Diisopropyl ketone (e) Acetone (f) 2,5-Dimethylcyclohexanonearrow_forward
- 17-47 What is the characteristic structural feature of a hemiacetal? Of an acetal?arrow_forward14-55 (Chemical Connections 140 The legal definition of being under the influence of alcohol is based on blood alcohol content. What is the relationship between breath alcohol content and blood alcohol content?arrow_forwardHydrolysis of an acetal in an acid solution would yield O a) an aldehyde, a ketone, and one alcohol b) an aldehyde, a ketone, and two alcohols c) an aldehyde or a ketone and one alcohol O d) an aldehyde or a ketone and two alcoholsarrow_forward
- The name of this organic compound. CH,CH,-C-CH,-CHO O 3-ketopentanal 3-oxobutanal 3-oxopentanal O 3-pentanone The type of this organic compound. Ochiral ketone symmetrical ketone achiral ketone unsymmetrical ketonearrow_forwardDefine carboxylationarrow_forwardAn alcohol is formed by oxidation of a ketone. True or falsearrow_forward
- Write names and formulas for simple ketones.arrow_forwardHydrolysis of an acetal in aqueous acid gives an aldehyde or a ketone and two molecules of alcohol or one molecule of a diol. Draw the structural formulas for the products of hydrolysis of the following acetals in aqueous acid.arrow_forwardTollens’ reagent test to identify aldehyde and ketones; Write whether the following aldehydes and ketones are positive or negative for the Tollens’ Test. Propionaldehyde (propanal) Benzaldehyde Methyl Ethyl ketone (Unknown) Acetophenonearrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning


