
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
9th Edition
ISBN: 9781292151229
Author: Wade, LeRoy G.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 20.36SP
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
- a. trans – 1 – bromobut - 2 – ene → trans - pent – 3 – enoic acid (two ways)
- b. hex – 3 – ene → propanoic acid
- c. but – 2 – enal → but – 2 – enoic acid
- d. hexanoicacid → hexanal
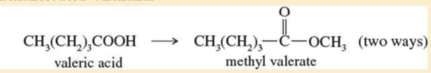
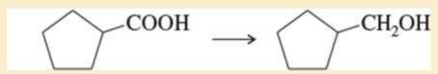
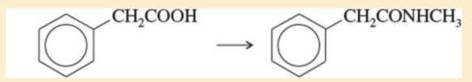
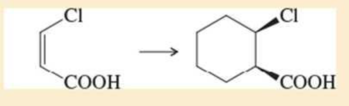
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the organic product(s) formed when CH3CH₂CH₂OH is treated with each reagent.
a. H₂SO4
d. HBr
g. TsCl, pyridine
b. NaH
h. [1] NaH; [2] CH₂CH₂Br
e. SOCI₂, pyridine
f. PBr3
c. HCI + ZnCl₂
Hint: NaH deprotonates the alcohol forming an alkoxide
Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent.a. HClb. CH3COClc. (CH3CO)2Od. excess CH3Ie. (CH3)2C = Of. CH3COCl, AlCl3g. CH3CO2Hh. NaNO2, HCli. Part (b), then CH3COCl, AlCl3j. CH3CHO, NaBH3CN
Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent.
a.H2SO4
b.NaH
c.HCl + ZnCl2
d.HBr
e.SOCl2, pyridine
f.PBr3
g.TsCl, pyridine
h. [1] NaH; [2] CH3CH2Br [1]
i.TsCl, pyridine; [2] NaSH
j.POCl3, pyridine
Chapter 20 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
Ch. 20.2C - Prob. 20.1PCh. 20.2C - Name the following carboxylic acids (when...Ch. 20.4B - Rank the compounds in each set in order of...Ch. 20.5 - Prob. 20.4PCh. 20.5 - Phenols are less acidic than carboxylic acids,...Ch. 20.5 - Prob. 20.6PCh. 20.7A - Prob. 20.7PCh. 20.7B - Prob. 20.8PCh. 20.7D - Draw all four resonance forms of the fragment at...Ch. 20.7D - a. Why do most long-chain fatty acids show a large...
Ch. 20.10 - Prob. 20.13PCh. 20.10 - A carboxylic acid has two oxygen atoms, each with...Ch. 20.10 - Prob. 20.15PCh. 20.10 - The mechanism of the Fischer esterification was...Ch. 20.10 - Prob. 20.17PCh. 20.12 - Show how to synthesize the following compounds,...Ch. 20.13 - Show how you would synthesize the following...Ch. 20.14 - Prob. 20.20PCh. 20.14 - Prob. 20.21PCh. 20.15 - Propose a mechanism for the reaction of benzoic...Ch. 20.15 - Prob. 20.23PCh. 20.15 - Prob. 20.24PCh. 20 - Prob. 20.25SPCh. 20 - Give both IUPAC names and common names for the...Ch. 20 - Draw the structures of the following compounds. a....Ch. 20 - Prob. 20.28SPCh. 20 - Arrange each group of compounds in order of...Ch. 20 - Predict the products (if any) of the following...Ch. 20 - Rank the following isomers in order of increasing...Ch. 20 - Prob. 20.32SPCh. 20 - What do the following pKa values tell you about...Ch. 20 - Given the structure of ascorbic acid (vitamin C):...Ch. 20 - Prob. 20.35SPCh. 20 - Show how you would accomplish the following...Ch. 20 - Predict the products and propose mechanisms for...Ch. 20 - Prob. 20.38SPCh. 20 - Prob. 20.39SPCh. 20 - Prob. 20.40SPCh. 20 - Prob. 20.44SPCh. 20 - Prob. 20.45SPCh. 20 - Predict the major form of each compound when it is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 4. WHAT ARE IMINES? HOW ARE THEY FORMED? 5. WHAT IS GRIGNARD REACTION? WHAT CONSTITUTES A GRIGNARD REAGENT? 6. WHAT IS KETO-ENOL TAUTOMERIZATION?arrow_forward1. What is the product of the following reaction? سد A. B. HO [H+] -H₂O (Η کھے D. HO Oarrow_forward1. Write the structure of the major organic product for reactions a, b and e? H₂C b. H₂c- CH, CH₂ Cold KMnO, OH A KMnO, H c. CH3CH₂-CH=CH-CH₂CH3 + H₂SO4 →arrow_forward
- Draw the product formed when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents, no reaction occurs. a. NaHCO3 b. NaOH c. SOCl2 d. NaCl e. NH3(1equiv) f. NH3, ∆ g. CH3OH, H2SO4 h. CH3OH, −OH i. [1] NaOH; [2] CH3COCl j. CH3NH2, DCC k. [1] SOCl2; [2] CH3CH2CH2NH2 (excess) l. [1] SOCl2; [2] (CH3)2; [2] (CHarrow_forwardWhat explains why many aldehydes and ketones can undergo self- condensation reactions in basic conditions? A. The alpha carbon can lose a proton and act like a nucleophile and the carbonyl carbon a an electrophile B. The alpha carbon can gain a proton and act like an electrophile and the carbonyl carbon is a nucleophile C. The oxygen of the carbonyl group can attack the carbon of the carbonyl group D. Only esters can undergo self-condensation reactionsarrow_forward10. Why do aldehydes undergo nucleophilic addition reactions (rxns from Unit 10) while esters undergo nucleophilic acyl substitution (rxns from Unit 11) reactions? A) The carbonyl carbon of an ester is more electrophilic than that of an aldehyde. B) Aldehydes are more sterically hindered than esters. C) Once the nucleophile adds to an aldehyde, the tetrahedral intermediate is too sterically hindered to eliminate one of the attached groups. D) The ester carbonyl carbon is sp3 hybridized while the aldehyde carbonyl carbon is sp2 hybridized. E) Once the nucleophile adds to an aldehyde, neither H- nor R- can be eliminated since they are strongly basic.arrow_forward
- e) sec-butyl bromide • Question 2: Identify the nucleophile and leaving group and draw the products of each reaction. a. b. Br + + OCH₂CH3 NaOH d. Br + N3 + NaCNarrow_forwardIdentify the role/function of the indicated molecule in the reaction? 1. LIAIH, ether 2. Но NaN3 Br -NH2 a. Nucleophile b. Electrophile c. Base O d. Acidarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. 1. LIAIH4 2. H₂O CI Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License