
Inorganic Chemistry
5th Edition
ISBN: 9780321811059
Author: Gary L. Miessler, Paul J. Fischer, Donald A. Tarr
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 3.19P
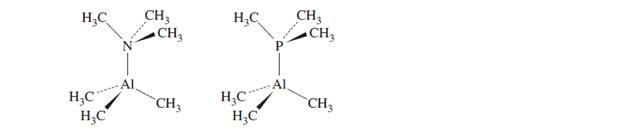
Make the following comparisons about the molecules shown next, and briefly explain yourchoices.
a. Which molecule has the smaller
b. Which molecule has the smaller
c. Which molecule has the longer
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the compound drawn below, decide which statement is true.
OH
It contains a sp hybridized atom.
It is more soluble in CHCI3 than in H20.
a
b
Its boiling point is lower than cyclohexane.
The oxygen atom has trigonal planar geometry.
It is more soluble in H20 than in CHCI3.
d.
O0000
How are I and II related?
I
and
H3C-C=CH
11
Select one:
O A. They are identical compounds.
OB. They are different compounds, not isomers.
OC. They are resonance structures.
O D. They are isomers.
Which of the follow
0
hn. Ch3
oh. What is this formula molecular structure, number of carbon sites how many hydrogen sites how many atoms each How many atoms in each type of atom in the structure give a molecular formula and identify tw functional gr
Chapter 3 Solutions
Inorganic Chemistry
Ch. 3.1 - Use electron-dot diagrams and formal charges to...Ch. 3.2 - Predict the structures of the following ions....Ch. 3.2 - Prob. 3.3ECh. 3.2 - Which molecule has the smallest bond angle in each...Ch. 3.2 - Briefly account for the following observations: a....Ch. 3.2 - Does this approach work for different steric...Ch. 3 - The dimethyldithiocarbamate ion, [S2CN( CH 3 )2]-...Ch. 3 - Several resonance structures are possible for each...Ch. 3 - Draw the resonance structures for the...Ch. 3 - Three isomers having the formula N2CO are known:...
Ch. 3 - Show the possible resonance structures for nitrous...Ch. 3 - Nitric acid, which exists as HNO3 molecules in the...Ch. 3 - L. C. Allen has suggested that a more meaningful...Ch. 3 - Give Lewis dot structures and sketch the shapes of...Ch. 3 - Give Lewis dot structures and sketch the shapes of...Ch. 3 - Give Lewis dot structures and sketch the shapes of...Ch. 3 - Explain the trends in bond angles and bond lengths...Ch. 3 - Select from each set the molecule or ¡on having...Ch. 3 - a. Compare the structures of the azide ion, N3 ,...Ch. 3 - Consider the series OCl2,O( CH3)2 , and O( SiH3)2...Ch. 3 - Two ions isoelectronic with carbon suboxide, C3O2...Ch. 3 - Explain the following: a. Ethylene, C2H4 , is a...Ch. 3 - Explain the following: a. PCI5 is a stable...Ch. 3 - X-ray crystal structures of ClOF3 and BrOF3 have...Ch. 3 - Make the following comparisons about the molecules...Ch. 3 - Prob. 3.20PCh. 3 - A solution containing the lO2F2 ion reacts slowly...Ch. 3 - The XeOF3 anion has been reported recently (D. S....Ch. 3 - Predict the structure of l(CF3)Cl2 . Do you expect...Ch. 3 - a. Which has the longer axial PF distance, PF2(...Ch. 3 - Prob. 3.25PCh. 3 - SeCl62,TeCl62 , and CIF6 are all octahedral, but...Ch. 3 - Prob. 3.27PCh. 3 - The thiazyldichloride ion, NSCl2- , is...Ch. 3 - Sketch the most likely structure of PCl3Br2 and...Ch. 3 - a. Are the CF3 groups in PCl3( CF3)2 more likely...Ch. 3 - Of the molecules C1SO2CH3,C1SO2CF3 , and ClSO2CCl3...Ch. 3 - Prob. 3.32PCh. 3 - Prob. 3.33PCh. 3 - Prob. 3.34PCh. 3 - Prob. 3.35PCh. 3 - Although the CF distances and the FCF bond angles...Ch. 3 - The Cl...Cl distance in CCl4 is 289 pm, and the...Ch. 3 - The FCF angle in F2CO , shown here, is 109.5°; the...Ch. 3 - Compounds in which hydrogen is the outer atom can...Ch. 3 - For each of the following bonds, indicate which...Ch. 3 - Give Lewis dot structures and shapes for the...Ch. 3 - Give Lewis dot structures and sketch the shapes...Ch. 3 - Which of the molecules in Problem 3.41 are polar?Ch. 3 - Which of the molecules in Problem 3.42 are polar?Ch. 3 - Prob. 3.45PCh. 3 - Prob. 3.46P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the compounds in the Figure below can have cis and trans isomers? B A D CH3 CH,-CH=C-CH-CH, CH3 A. Compound B B. Compounds A, B and D C. Compound A D. Compounds A and Barrow_forward1. How many electrons does carbon have in its valence shell? 2. Carbon most frequently forms bonds with 3. Draw methane (CH4) using electron distribution diagrams. Example: Neon (₁oNe) 4. 5. Label the isomers: H HC H HH HH CH OH Hydroxyl H H H H H HH H C-H H are organic molecules that only consist of carbon and hydrogen. H c-c 6. Draw a line to connect the functional group to its structure: H SH с-с H - OH WA A -N H H Carbonyl Carboxyl Amino Sulfhydry! 11 Phosphate -P-0 0- Methyl andarrow_forward15. a. Draw two constitutional isomers with the formula CaHsBrCl ( Isomer 1 Isomer 2 b. Describe ALL the intermolecular forces isomer 1 can participate in 16. For the molecule below, OH (a) Circle all functional groups present. (b) Label two of the functional groups next to where you circled them 3arrow_forward
- Which of the organic compounds shown below exhibit cis-trans isomerism? Select one: a. CH3CH=CHCH2CH3 O b. CH2=CH(CH3) O c. CH2=C(CH3)2 O d. CH2=C(CI)2arrow_forward11:51 l 5G Question 9 of 25 Submit 1. O3 2. CH3SCH3 Select to Draw CSH100 Select to Draw CH2Oarrow_forwardCO₂H -OH -OH heat 1. SOCI 2, A 2. NaO,CCH2CH3 ?arrow_forward
- For the 6-carbon molecule below, how would you expect it to break into two pieces? two 3 carbon units one 2 carbon unit and one 4 carbon unit one 1 carbon unit, one 2 carbon unit, and one 3 carbon unit one 1 carbon unit and one 5 carbon unitarrow_forwardQUESTION 4: Fill in the blanks if you dare! неја OH H₂C 1) NaBH4 2) SOCIz OH OH CH 3arrow_forwardWhat is a hydrocarbon? What is the difference between a saturated hydrocarbon and an unsaturated hydrocarbon? Distinguish between normal and branched hydrocarbons. What is an alkane? What is a cyclic alkane? What are the two general formulas for alkanes? What is the hybridization of carbon atoms in alkanes? What are the bond angles in alkanes? Why are cyclopropane and cyclobutane so reactive? The normal (unbranched) hydrocarbons are often referred to as straight-chain hydrocarbons. What does this name refer to? Does it mean that the carbon atoms in a straight-chain hydrocarbon really have a linear arrangement? Explain. In the shorthand notation for cyclic alkanes, the hydrogens are usually omitted. How do you determine the number of hydrogens bonded to each carbon in a ring structure?arrow_forward
- What is the meaning of the term tertiary (3) when it is used to classify alcohols? Draw a structural formula for the one tertiary (3) alcohol with the molecular formula C4H10O.arrow_forwardModule 11 HW Chapter 20 - Selected Exercises Question 4 of 8 Determine the structures of compounds A through F: HO. Na,Cr0, SOCI, xs NH3 C A H,SO H,O [H*] 1) LIAI(OR);H 2) H30+ ELOH Farrow_forwardConsider the structure of ascorbic acid below. НО 5 3 HO. O HỞ `H i. C5 is • hybridized. ii. How many C-C sigma bonds are there in the structure of ascorbic acid? iii. is the C-C single bond with the highest bond dissociation energy.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY