
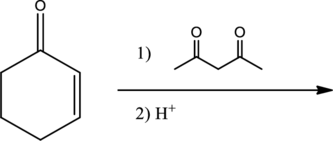
Interpretation:
For the given reaction, whether an efficient Michael reaction is expected or not has to be determined.
Concept Introduction:
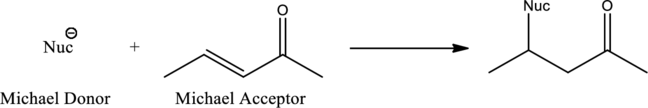
Michael reaction is the reaction between a Michael donor which acts as a nucleophile and a Michael acceptor. Important condition for the Michael reaction to occur is the formation of stabilized nucleophile. 1,4-addition is known as Michael addition where the beta-keto compound acts as nucleophile. General scheme for Michael reaction can be given as,
Explanation of Solution
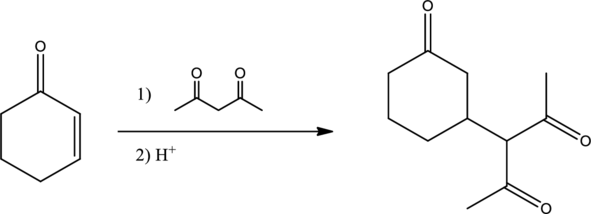
Given reaction is,
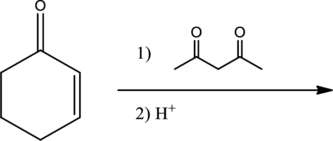
From the above reaction, it is found that there is an alpha,beta-unsaturated
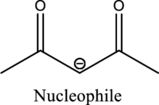
Therefore, the given reaction will be an efficient Michael reaction. It can be represented as shown below,
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
- Draw ALL the major product obtained when the following alkyl halide undergoes an E1 reaction. The E1 reaction below may have more than one product that forms (less substituted, more substituted, E/Z). Be sure to draw out all of the products that form.arrow_forwardThere are three possible products for the E1 elimination of this reaction. Draw one of the major products and the minor product. Ignore any inorganic byproducts. H3O+ heat OH Draw E1 Major Product + Draw Minor E1 Productarrow_forwardAnalyze the reaction schemes below and state (with reasons) whether the reaction conditions supplied in each reaction scheme will result in the products shown as MAJOR PRODUCTS. If wrong products are shown, draw the correct products and provide mechanistic details (curly arrows) of how the right (major) products were formed.arrow_forward
- Choose the correct set of reagents that would lead to this major product under these conditions.arrow_forwardConsider this two-step synthesis. Show how compound on the right can be synthesized by providing reagents for each step and the product in the first synthetic step (use the boxes below). step 1 step 2 Br NO Briefly describe the strategy you outlined above that will ensure a stereochemically pure product (not a racemic mixture).arrow_forwardDraw Zaitsev and Hofmann products that are expected when each of the following compounds is treated with a strong base to give an E2 ELIMINATION product. Hint: Some compounds cannot produce both elimination products because they do not have two beta-hydrogen atoms: in these cases only ONE product will be possible. In cases when neither elimination products are possible, there will be NO REACTION. ? O A O O O B U Br t Zaitsev Hofmann only ONE product is possible B only ONE product is possible Hofmann Zaitsevarrow_forward
- Answer the question below the reaction. ta The reaction above proceeds through which type of mechanism? SN2 SN1 E1 E2 OH + Excess NH4C1 H₂SO4 + H₂Oarrow_forwardDraw Zaitsev and Hofmann products that are expected when each of the following compounds is treated with a strong base to give an E2 ELIMINATION product. Hint: Some compounds cannot produce both elimination products because they do not have two beta-hydrogen atoms: in these cases only ONE product will be possible. In cases when neither elimination products are possible, there will be NO REACTION. t ? A C O O O A B U xx Zaitsev Hofmann only ONE product is possible B D NO products are possible (NO REACTION) Hofmann dd Zaitsevarrow_forwardGive the major product(s) for each step of the following sequence of reactionsarrow_forward
- SN1 reactions undergo carbocation rearrangements, but E1 reactions do not because the carbocation intermediate does not last as long during the elimination process. True O Falsearrow_forwardClassify the following reaction as an Sn1, Sn2, E1, E1cB, E2 reaction. Can you show me the leaving group and explain how to find a leaving group? And how to identify the other reactions?arrow_forwardConsider the following reaction scheme (note that the reagent shown above the arrow is DBN"). Draw in the expected major product AND indicate what mechanism the reaction will follow. Product: Mechanism:arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





