
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 8.9, Problem 8.72P
Interpretation Introduction
Interpretation:
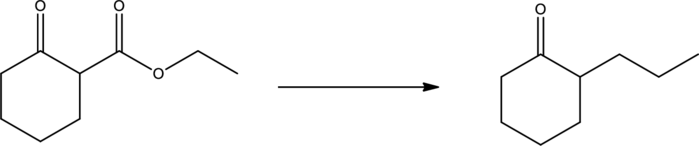
Synthesis has to be proposed for the given transformation.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
propose a mechanism for the following transformations
Rimantadine is an antiviral drug used to treat people infected with life-threatening influenza viruses. Identify the starting ketone that
would be necessary in order to prepare rimantadine via a reductive amination.
NH2
Rimantadine
With the starting material provided, propose a synthesis pathway to prepare the diketone shown below.
سلام
Chapter 8 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.2 - Prob. 8.9PCh. 8.2 - Prob. 8.10PCh. 8.2 - Prob. 8.11PCh. 8.2 - Prob. 8.12P
Ch. 8.2 - Prob. 8.13PCh. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.4 - Prob. 8.20PCh. 8.4 - Prob. 8.21PCh. 8.4 - Prob. 8.22PCh. 8.4 - Prob. 8.23PCh. 8.5 - Prob. 8.25PCh. 8.5 - Prob. 8.26PCh. 8.5 - On a separate piece of paper, draw a mechanism for...Ch. 8.6 - Prob. 8.29PCh. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Prob. 8.42PCh. 8.7 - Prob. 8.43PCh. 8.7 - Prob. 8.44PCh. 8.7 - Prob. 8.45PCh. 8.7 - Prob. 8.47PCh. 8.7 - Prob. 8.48PCh. 8.7 - Prob. 8.49PCh. 8.7 - Prob. 8.50PCh. 8.8 - Prob. 8.52PCh. 8.8 - Prob. 8.53PCh. 8.8 - Prob. 8.54PCh. 8.8 - Prob. 8.55PCh. 8.8 - Prob. 8.57PCh. 8.8 - Prob. 8.58PCh. 8.8 - Prob. 8.59PCh. 8.8 - Prob. 8.60PCh. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Prob. 8.64PCh. 8.9 - Prob. 8.66PCh. 8.9 - Prob. 8.67PCh. 8.9 - Prob. 8.68PCh. 8.9 - Prob. 8.69PCh. 8.9 - Prob. 8.70PCh. 8.9 - Prob. 8.71PCh. 8.9 - Prob. 8.72PCh. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.10 - Prob. 8.78PCh. 8.10 - Prob. 8.79PCh. 8.10 - Prob. 8.80PCh. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...
Knowledge Booster
Similar questions
- A) Propose an efficient synthesis for the following transformation: + en B) Propose an efficient synthesis for the following transformation: он + enarrow_forwardpropose a mechanism for the following transformationarrow_forwardPropose preparative routes to the following ketones using an enamine intermediate - you must make the bond in red.arrow_forward
- 1. What reaction involving amines is required to produce N-Propylcycloheptanamine? Propose a synthesis and mechanism2. Is the reaction below favorable or unfavorable? State your reasons.arrow_forwardPropose a synthesis for the following transformation: HOarrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrro- lidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place. Me Me Но N: 'N' AcO Ме pyrrolidinium acetate Me Me Mearrow_forward
- Following are two possible retrosynthetic analyses for the anticholinergic drug cycri- mine. Fill in the details of each potential synthesis. O: OH + H. Cycrimine (гасemic)arrow_forwardThe ketone whose 1H NMR spectrum is shown here was obtained as the product of an acetoacetic ester synthesis. What alkyl halide was used in the synthesis?arrow_forwardPropose a synthesis for the following transformation:arrow_forward
- Ethanolamine ammonia lyase, a coenzyme B12–requiring enzyme, catalyzes the following reaction. Propose a mechanism for this reaction.arrow_forwardIn the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals inlarge doses, paraldehyde is used as a sedative or hypnotic. Propose a mechanism for the formation of paraldehyde.arrow_forwardPropose a synthesis for the following transformation. OEt HO,arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning