
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 8.2, Problem 8.13P
Interpretation Introduction
Interpretation:
Mechanism has to be proposed for the given transformation.
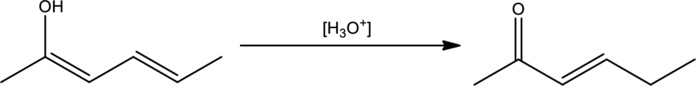
Concept Introduction:
In order to draw mechanism for a keto‑enol tautomerism, three steps has to be followed. They are,
- Order that has to be used. Whether to protonate or deprotonate first.
- The site for protonation or deprotonation.
- Reagents to be used when drawing the proton transfer steps.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Show two reaction schemes to synthesize the following compounds. Pick one reactionscheme and draw the step-by-step reaction mechanism
Use the reaction given below to answer the following questions.
A
CH₂OH
B
H₂O+
C
+ CH3NH3*
D
a) What are the approximate pKas of compounds A and B?
b) The reaction given above is irreversible. Give a detailed explanation of the reaction and reaction
mechanism. Your explanation should indicate your knowledge of the reactivity of carboxylic acid
derivatives.
Propose a mechanism for the reaction of benzyl acetate with methylamine. Label theattacking nucleophile and the leaving group, and draw the transition state in which theleaving group leaves.
Chapter 8 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.2 - Prob. 8.9PCh. 8.2 - Prob. 8.10PCh. 8.2 - Prob. 8.11PCh. 8.2 - Prob. 8.12P
Ch. 8.2 - Prob. 8.13PCh. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.4 - Prob. 8.20PCh. 8.4 - Prob. 8.21PCh. 8.4 - Prob. 8.22PCh. 8.4 - Prob. 8.23PCh. 8.5 - Prob. 8.25PCh. 8.5 - Prob. 8.26PCh. 8.5 - On a separate piece of paper, draw a mechanism for...Ch. 8.6 - Prob. 8.29PCh. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Prob. 8.42PCh. 8.7 - Prob. 8.43PCh. 8.7 - Prob. 8.44PCh. 8.7 - Prob. 8.45PCh. 8.7 - Prob. 8.47PCh. 8.7 - Prob. 8.48PCh. 8.7 - Prob. 8.49PCh. 8.7 - Prob. 8.50PCh. 8.8 - Prob. 8.52PCh. 8.8 - Prob. 8.53PCh. 8.8 - Prob. 8.54PCh. 8.8 - Prob. 8.55PCh. 8.8 - Prob. 8.57PCh. 8.8 - Prob. 8.58PCh. 8.8 - Prob. 8.59PCh. 8.8 - Prob. 8.60PCh. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Prob. 8.64PCh. 8.9 - Prob. 8.66PCh. 8.9 - Prob. 8.67PCh. 8.9 - Prob. 8.68PCh. 8.9 - Prob. 8.69PCh. 8.9 - Prob. 8.70PCh. 8.9 - Prob. 8.71PCh. 8.9 - Prob. 8.72PCh. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.10 - Prob. 8.78PCh. 8.10 - Prob. 8.79PCh. 8.10 - Prob. 8.80PCh. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...
Knowledge Booster
Similar questions
- Minoxidil is a molecule that causes hair growth in some people. It was originally synthesized as a vasodilator for the treatment of hypertension (high blood pressure). Most of the patients taking the drug for hypertension were seen to grow body hair. Due to other side effects, its oral use was stopped, but it became popular as a topical cream to promote hair growth. The first key reaction in one synthesis of minoxidil follows. Draw the product of this reaction.arrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forward1) Show how the below transformations have occurred through the alkylation of imine and hydrazone intermediates respectively. Show all the reagents and intermediate compounds (the amount of arrows do not indicate the specific number of steps required) a) b) Imine intermediate Hydrazone intermediate OH gifarrow_forward
- Complete the following reaction of hydrolysis using arrows and drawing the intermediate products Catión estabilizado por resonancia Catión oxonio MeO OMe - MeOH R R Acetal Catión oxonio Catión oxonio Meo OH R- R Hemiacetal MeOH Catión estabilizado por resonancia -H® R R Compuesto carbonilicoarrow_forwardThe following reaction takes place in multiple steps, with a vital intermediate isolated after the treatment with ammonia.arrow_forwardPropose an appropriate intermediate for the following reactionarrow_forward
- I am unsure about how to draw out all the steps for this synthesis reaction.arrow_forwardPredict the major product(s) that are expected when the following compound is heated with concentrated HBr. Modify the given drawing of the starting material to draw only the organic product(s). CH3 Edit Drawingarrow_forwardWhich reagents will give the following synthesis reaction?arrow_forward
- How many elimination products will be formed in the following reaction?arrow_forwardStep 1 X Incorrect. Which of the mechanistic steps is shown in this step of the mechanism? Draw step 1 of the mechanism. H;C CH3 ČH3 Edit Drawingarrow_forwardhow would a mechanism for this reaction proceed ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning