
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.36P
The purine heterocycle occurs commonly in the structure of DNA.
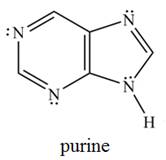
a. How is each
b. In what type of orbital does each lone pair on a
c. How many
d. Why is purine aromatic?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a.How many π electrons does C contain?
b.How many π electrons are delocalized in the ring?
c.Explain why C is aromatic.
Which molecules below are anti- aromatic?
D
A.
Structure D
Structure B
Structure A
Structure C
B.
C.
D.
Which molecules below are aromatic?
A.
B.
Structure D
Structure B
Structure A
Structure C
Structure E
D.
E.
Chapter 17 Solutions
Organic Chemistry
Ch. 17 - Prob. 17.1PCh. 17 - Problem 17.2 What orbitals are used to form the...Ch. 17 - Problem-17.3. Give the IUPAC name for each...Ch. 17 - Prob. 17.4PCh. 17 - Problem-17.5 What is the structure of propofol,...Ch. 17 - Problem 17.6 What is the structure of a compound...Ch. 17 - Problem 17.7 How many NMR signals does each...Ch. 17 - Prob. 17.8PCh. 17 - Prob. 17.9PCh. 17 - Prob. 17.10P
Ch. 17 - Prob. 17.11PCh. 17 - Prob. 17.12PCh. 17 - Prob. 17.13PCh. 17 - Problem 17.14 Januvia, the trade name for...Ch. 17 - Prob. 17.15PCh. 17 - Problem 17.16 Rank the following compounds in...Ch. 17 - Problem 17.17 Draw the seven resonance structures...Ch. 17 - Prob. 17.18PCh. 17 - Prob. 17.19PCh. 17 - Prob. 17.20PCh. 17 - Prob. 17.21PCh. 17 - Problem 17.22 How many NMR signals does ...Ch. 17 - 17.23 Name each compound and state how many lines...Ch. 17 - Prob. 17.24PCh. 17 - Prob. 17.25PCh. 17 - Prob. 17.26PCh. 17 - 17.27 Give the IUPAC name for each compounds.
a....Ch. 17 - 17.28 Draw a structure corresponding to each...Ch. 17 - 17.29 a. Draw the 14 constitutional isomers of...Ch. 17 - Prob. 17.30PCh. 17 - Prob. 17.31PCh. 17 - Prob. 17.32PCh. 17 - 17.33 Label each compound as aromatic,...Ch. 17 - Prob. 17.34PCh. 17 - 17.35 Pentalene, azulene, and heptalene are...Ch. 17 - 17.36 The purine heterocycle occurs commonly in...Ch. 17 - Prob. 17.37PCh. 17 - 17.38
How many electrons does C contain?
How...Ch. 17 - Prob. 17.39PCh. 17 - 17.40 Explain the observed rate of reactivity of...Ch. 17 - 17.41 Draw a stepwise mechanism for the following...Ch. 17 - Prob. 17.42PCh. 17 - 17.43 Draw additional resonance structures for...Ch. 17 - Prob. 17.44PCh. 17 - Prob. 17.45PCh. 17 - 17.46 Which compound in each pair is the stronger...Ch. 17 - 17.47 Treatment of indene with forms its...Ch. 17 - Prob. 17.48PCh. 17 - 17.49 Draw the conjugate bases of pyrrole and...Ch. 17 - 17.50 a. Explain why protonation of pyrrole occurs...Ch. 17 - Prob. 17.51PCh. 17 - Prob. 17.52PCh. 17 - 17.53 How many signals does each compound...Ch. 17 - 17.54 Which of the diethylbenzene isomers (ortho,...Ch. 17 - 17.55 Propose a structure consistent with each...Ch. 17 - 17.56 Propose a structure consistent with each...Ch. 17 - 17.57 Thymol (molecular formula ) is the major...Ch. 17 - 17.58 You have a sample of a compound of molecular...Ch. 17 - 17.59 Explain why tetrahydrofuran has a higher...Ch. 17 - 17.60 Rizatriptan (trade name Maxalt) is a...Ch. 17 - 17.61 Zolpidem (trade name Ambien) promotes the...Ch. 17 - 17.62 Answer the following questions about...Ch. 17 - 17.63 Stanozolol is an anabolic steroid that...Ch. 17 - Prob. 17.64PCh. 17 - 17.65 Use the observed data to decide whether C...Ch. 17 - Prob. 17.66PCh. 17 - Prob. 17.67PCh. 17 - Prob. 17.68PCh. 17 - 17.69 Although benzene itself absorbs at in its ...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which among the five aromatic molecules will have the least ability to undergo molecularstacking?A. Structure 1B. Structure 2C. Structure 3D. Structure Which aromatic structures contains functional groups which are electron-withdrawing?A. Structures 1, 2 and 4B. Structures 2, 3 and 5C. Structures 2, 4, and 5D. Structures 1, 3, and 4 Which aromatic structures contain functional groups which are electron-donating?A. Structures 1 and 3B. Structures 1 and 4C. Structures 3 and 5D. Structures 3 and 4 In which structure do you expect two products after alkylation?A. Structure 1B. Structure 2C. Structure 3D. Structure 4 In which carbon position will an incoming halogen be located in Structure 3, relative tothe OH group?A. Ortho positionB. Meta positionC. Para positionD. Ortho and para position In which aromatic molecule do you expect to have a carbocation form during resonancestabilization?A. Structures 2, 3 and 4B. Structures 2, 4 and 5C. Structures 2, 3 and 5D. Structures 1, 3 and 4arrow_forwardZolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia. a.In what type of orbital does the lone pair on each N atom in the heterocycle reside? b. Explain why the bicyclic ring system that contains both N atoms is aromatic. c.Draw all reasonable resonance structures for the bicyclic ring system.arrow_forward2. Which structure is NOT aromatic? a d. H3C :Br: DOarrow_forward
- Select all aromatic rings that contain deactivating groups. a. d. a b C P e f -ħ NH₂ b. NO₂ SO₂H C. LOCH3arrow_forwardWhich of the following compounds is aromatic? A. only A and B B. only A and C C. only B and C D.only Barrow_forwarda) Which molecule has a higher electron density in the aromatic ring? Explain. H₂C O CO₂H CO₂H H₂C-N CH3 CH3 b) which is more acidic?arrow_forward
- INSTRUCTIONS: Choose the letter of the BEST answer for each item. 1. How many lone pairs are involved in sustaining the conjugation of pyridine? A. One B. Two C. Three D. Four 2. Benzopyrene, naphthalene and pyrene are members of these group of aromatic compounds: A. Benzenoid aromatic compounds B. Non-benzenoid aromatic compounds C. Heterocyclic aromatic compounds D. Heteronuclear compounds 3. What type of aromatic compound is pyridine? A. Heterocyclic aromatic compound B. Benzenoid aromatic compound C. Non-benzenoid aromatic compound D. Homonuclear cyclic compound 4. What property of aromatic rings prevent the involvement of the conjugated structure to addition reactions? A. Radical stabilization B. Resonance stability C. Inductive effect D. Aromatic effect 5. Which electrophilic aromatic substitution reaction is described when aniline is transformed into para-aminotolouene? A. Nitration B. Friedel Crafts alkylation C. Oxidation D. Halogenation 6. What type of relationship does…arrow_forwardWhich base is at the 5’ end of the structure shown? A.) A B.) C C.) G D.) T E.) Uarrow_forwardWhich compound is not aromatic? A. only A and D B. A, B and C C. only B D. only Aarrow_forward
- Answer the following questions about erlotinib and terbinafine. Erlotinib, sold under the trade name Tarceva, was introduced in 2004 for the treatment of lung cancer. Terbinafine is an antifungal medication used to treat ringworm and fungal nail infections. a.Which C–H bond in erlotinib is most acidic? b.What orbitals are used to form the shortest C–C single bond in erlotinib? c.Rank the labeled bonds in terbinafine in order of increasing bond strength. d. Draw two additional resonance structures for terbinafine that contain all uncharged atoms.arrow_forwardC. D. O: :O: The lone pair in compound C is Compound C is In compound D, not aromatic. aromatic. delocalized. not delocalized. one lone pair is delocalized. both lone pairs are not delocalized. both lone pairs are delocalized. Compoundarrow_forward59. Bond Strength I. Cyclobutane II. Cyclopentane 60. Optical Rotation I. 70% R, 30% S 11.30% R, 70% S 61. Which of the following is/ are alternant aromatic compounds? A. benzene B. naphthalene C. anthracene D. all of the given choices 62. Which of the following is/ are not aromatic? A. Cyclobutadiene B. Cyclopentadiene C. Cyclohexadiene D. All of the given choices 63. The following can exhibit tautomerism EXCEPT: A. benzaldehyde B. ethanal C. Cyclohexane carbaldehyde D. Cyclohexanone 64. Which of the following substituents exhibits both - inductive effect and - Resonance effect when attached to the benzene ring? A. -OH B. -CI D. -NO2 D.-CH3 65. Alkyl groups if attached to halogen (example: tert-butyl bromide) would exhibit A. negative inductive effect B. positive inductive effect C. negative resonance effect D. positive resonance effect. 9arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY