
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.5, Problem 13P
Interpretation Introduction
a)
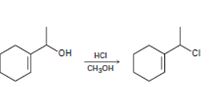
Interpretation:
The above substitution reaction belongs to SN1 type.
Concept introduction:
SN1 reaction occurs mostly in acidic condition. The OH group is first protonated by HCl. Spontaneous dissociation of the protonated alcohol occurs in a slow, rate determining step to yield a carbocation intermediate. The carbocation intermediate reacts with the chloride ion to yield the chloride product.
Interpretation Introduction
b)
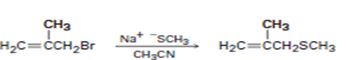
Interpretation:
The above substitution reaction belongs to SN2 type.
Concept introduction:
SN2 reaction occurs rapidly in
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Tell whether the following reaction is likely to be SN1, SN2, E1, E2 or E1cB, and predict the product.
A CHEM 245 student attempts the following reaction to synthesize the desired ether below,
but he is unsuccessful:
H2SO4, CH3OH
OCH3
desired ether
racemic mixture
(a) Use what you have learned in CHEM 241 to predict the structure of the major product he did collect:
H2SO4, CH3OH
(only one stereoisomer required)
(b) Propose a reaction sequence that would be more likely to provide him with the desired ether product:
OCH3
desired ether
racemic mixture
3.
Write out a complete mechanism for the reaction below:
CH3
H3C
CH3
H
H
Mechanism:
2.
Which of the following compounds is the most reactive towards electrophilic aromatic subsitition by Br2?
NO2
NO2
NO2
ČH3
NME2
NO2
A
В
C
E
A B C D w w
OO OOO O
Chapter 11 Solutions
Organic Chemistry
Ch. 11.1 - Prob. 1PCh. 11.2 - Prob. 2PCh. 11.2 - Prob. 3PCh. 11.3 - Prob. 4PCh. 11.3 - Prob. 5PCh. 11.3 - Rank the following compounds in order of their...Ch. 11.3 - Organic solvents like benzene, ether, and...Ch. 11.4 - Prob. 8PCh. 11.4 - Prob. 9PCh. 11.4 - Prob. 10P
Ch. 11.5 - Rank the following substances in order of their...Ch. 11.5 - 3-Bromo-1-butene and 1-bromo-2-butene undergo SN1...Ch. 11.5 - Prob. 13PCh. 11.6 - Review the mechanism of geraniol biosynthesis...Ch. 11.7 - Prob. 15PCh. 11.7 - What alkyl halides might the following alkenes...Ch. 11.8 - Prob. 17PCh. 11.8 - Prob. 18PCh. 11.9 - Prob. 19PCh. 11.12 - Prob. 20PCh. 11.SE - Prob. 21VCCh. 11.SE - From what alkyl bromide was the following alkyl...Ch. 11.SE - Prob. 23VCCh. 11.SE - Prob. 24VCCh. 11.SE - Prob. 25MPCh. 11.SE - Prob. 26MPCh. 11.SE - Prob. 27MPCh. 11.SE - Prob. 28MPCh. 11.SE - Prob. 29MPCh. 11.SE - Prob. 30MPCh. 11.SE - Prob. 31MPCh. 11.SE - Prob. 32MPCh. 11.SE - Metabolism of S-adenosylhomocysteine (Section...Ch. 11.SE - Reaction of iodoethane with CN- yields a small...Ch. 11.SE - One step in the urea cycle for ridding the body of...Ch. 11.SE - Prob. 36MPCh. 11.SE - Prob. 37MPCh. 11.SE - Propose a mechanism for the following reaction, an...Ch. 11.SE - Prob. 39APCh. 11.SE - The following Walden cycle has been carried out....Ch. 11.SE - Prob. 41APCh. 11.SE - Which reactant in each of the following pairs is...Ch. 11.SE - Prob. 43APCh. 11.SE - Prob. 44APCh. 11.SE - Prob. 45APCh. 11.SE - Prob. 46APCh. 11.SE - Prob. 47APCh. 11.SE - Prob. 48APCh. 11.SE - Propose structures for compounds that fit the...Ch. 11.SE - What products would you expect from the reaction...Ch. 11.SE - Prob. 51APCh. 11.SE - Prob. 52APCh. 11.SE - Prob. 53APCh. 11.SE - Prob. 54APCh. 11.SE - Prob. 55APCh. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Prob. 58APCh. 11.SE - Prob. 59APCh. 11.SE - Ethers can often be prepared by SN2 reaction of...Ch. 11.SE - Show the stereochemistry of the epoxide (see...Ch. 11.SE - Prob. 62APCh. 11.SE - In addition to not undergoing substitution...Ch. 11.SE - The tosylate of (2R, 3S)-3-phenyl-2-butanol...Ch. 11.SE - Prob. 65APCh. 11.SE - Prob. 66APCh. 11.SE - Prob. 67APCh. 11.SE - Prob. 68APCh. 11.SE - Prob. 69APCh. 11.SE - (S)-2-Butanol slowly racemizes on standing in...Ch. 11.SE - Reaction of HBr with (R)-3-methyl-3-hexanol leads...Ch. 11.SE - Treatment of 1-bromo-2-deuterio-2-phenylethane...Ch. 11.SE - Prob. 73APCh. 11.SE - Prob. 74APCh. 11.SE - In light of your answer to Problem 11-74, explain...Ch. 11.SE - Prob. 76APCh. 11.SE - Compound X is optically inactive and has the...Ch. 11.SE - When a primary alcohol is treated with...Ch. 11.SE - Prob. 79APCh. 11.SE - Amines are converted into alkenes by a two-step...Ch. 11.SE - The antipsychotic drug flupentixol is prepared by...
Knowledge Booster
Similar questions
- 7) Tell whether the following reaction is likely to be SN1, SN2, E1, E1cB, or E2, and predict the product. бол -CH₂ CH₂ COM КОН CH₂CH₂CHCH₂CH₂ Ethanolarrow_forwardWhich of the following compounds is the most reactive towards electrophilic aromatic subsitition by Br2? NO2 NO2 NO2 CH3 NMe2 NO2 В D E F A C AB C DEE O O O O OOarrow_forwardFor each reaction below, provide the structural formula for the reactants and the predicted products. Indicate which mechanism the reaction will follow and use curved arrow formalism to illustrate the predicted reaction mechanism. t-butylalcohol + NaBr ?arrow_forward
- Which alkyl halide below is most likely to undergo a hydride shift in an S№1 substitution reaction? Br 66x6 Br B C Br XO B - D Brarrow_forwardWhich of the reactions below are correctly identified? These are SN2 reactions:arrow_forwardPredict the products of the following substitution reactions, including correct stereochemistry. Label each as SN2 or SN1. DMF Br NaCN CH,OH acetone КОН CH3 Br CH3CH2OH ČH3arrow_forward
- Which of the following is the most likely structure of the following cation after rearrangement? II II IV Varrow_forwardHow many different carbocations will form from an SN1 ionization mechanism on the molecule below. Include all possible resonance forms even if a minor contributor. Br 0000 A W N 3 4arrow_forwardIn light of your answer to Problem 11-74, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, and why?arrow_forward
- Discuss the following nucleophilic substitutions in terms of reaction conditions and stereochemical output. RO. NaCN Br R= H, CH3 Identify the preferred substitution mechanism for the following reactions. CH3 RSH ROH R. X = CI, Br, I R= H, CGH5arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs via a nucleophilic substitution pathway. The reaction is 100% stereospecific.arrow_forwardSN1 SN2 E1 E2 Mechanisms Predict the major products or provide the starting material for the following reactionsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
