
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.SE, Problem 37MP
Interpretation Introduction
Interpretation:
The mechanism has to be proposed for the given synthesis.
Concept introduction:
Substitution reaction:
Alcohol is reaction with tosyl chloride in pyridine which provides retention of configuration of tosylated compound.
This is shown below,
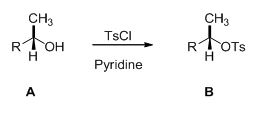
Given information:
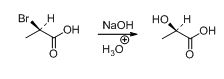
The given compound is shown below,
Answer:
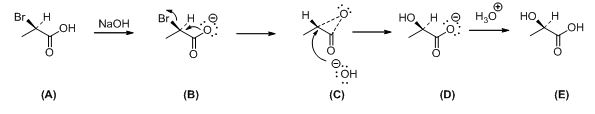
The mechanism of the reaction is shown below,
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
When an unsymmetrical Alkenes such as propane is treated with N-bromosuccinimide in aqueous dimethyl sulfoxide, the major product has the bromine atom bonded to the less highly substituted carbon atom. Is this Markovnikov or non-Markovnikov orientation? Show its complete reaction mechanism.
The reactions shown below are substitution reactions. Please state which mechanism they follow, and draw the mechanism of the reaction using mechanistic arrows
Provide the reaction (reactants -> products) with detailed mechanism for the following, with AlCl3 as catalyst. Indicate if the mechanism is substitution, addition of deletion.
Toluene + CHCl3
Benzene + CHCl3
1-Hexyne + CHCl3
3-Hexyne + CHCl3
Chapter 11 Solutions
Organic Chemistry
Ch. 11.1 - Prob. 1PCh. 11.2 - Prob. 2PCh. 11.2 - Prob. 3PCh. 11.3 - Prob. 4PCh. 11.3 - Prob. 5PCh. 11.3 - Rank the following compounds in order of their...Ch. 11.3 - Organic solvents like benzene, ether, and...Ch. 11.4 - Prob. 8PCh. 11.4 - Prob. 9PCh. 11.4 - Prob. 10P
Ch. 11.5 - Rank the following substances in order of their...Ch. 11.5 - 3-Bromo-1-butene and 1-bromo-2-butene undergo SN1...Ch. 11.5 - Prob. 13PCh. 11.6 - Review the mechanism of geraniol biosynthesis...Ch. 11.7 - Prob. 15PCh. 11.7 - What alkyl halides might the following alkenes...Ch. 11.8 - Prob. 17PCh. 11.8 - Prob. 18PCh. 11.9 - Prob. 19PCh. 11.12 - Prob. 20PCh. 11.SE - Prob. 21VCCh. 11.SE - From what alkyl bromide was the following alkyl...Ch. 11.SE - Prob. 23VCCh. 11.SE - Prob. 24VCCh. 11.SE - Prob. 25MPCh. 11.SE - Prob. 26MPCh. 11.SE - Prob. 27MPCh. 11.SE - Prob. 28MPCh. 11.SE - Prob. 29MPCh. 11.SE - Prob. 30MPCh. 11.SE - Prob. 31MPCh. 11.SE - Prob. 32MPCh. 11.SE - Metabolism of S-adenosylhomocysteine (Section...Ch. 11.SE - Reaction of iodoethane with CN- yields a small...Ch. 11.SE - One step in the urea cycle for ridding the body of...Ch. 11.SE - Prob. 36MPCh. 11.SE - Prob. 37MPCh. 11.SE - Propose a mechanism for the following reaction, an...Ch. 11.SE - Prob. 39APCh. 11.SE - The following Walden cycle has been carried out....Ch. 11.SE - Prob. 41APCh. 11.SE - Which reactant in each of the following pairs is...Ch. 11.SE - Prob. 43APCh. 11.SE - Prob. 44APCh. 11.SE - Prob. 45APCh. 11.SE - Prob. 46APCh. 11.SE - Prob. 47APCh. 11.SE - Prob. 48APCh. 11.SE - Propose structures for compounds that fit the...Ch. 11.SE - What products would you expect from the reaction...Ch. 11.SE - Prob. 51APCh. 11.SE - Prob. 52APCh. 11.SE - Prob. 53APCh. 11.SE - Prob. 54APCh. 11.SE - Prob. 55APCh. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Order each of the following sets of compounds with...Ch. 11.SE - Prob. 58APCh. 11.SE - Prob. 59APCh. 11.SE - Ethers can often be prepared by SN2 reaction of...Ch. 11.SE - Show the stereochemistry of the epoxide (see...Ch. 11.SE - Prob. 62APCh. 11.SE - In addition to not undergoing substitution...Ch. 11.SE - The tosylate of (2R, 3S)-3-phenyl-2-butanol...Ch. 11.SE - Prob. 65APCh. 11.SE - Prob. 66APCh. 11.SE - Prob. 67APCh. 11.SE - Prob. 68APCh. 11.SE - Prob. 69APCh. 11.SE - (S)-2-Butanol slowly racemizes on standing in...Ch. 11.SE - Reaction of HBr with (R)-3-methyl-3-hexanol leads...Ch. 11.SE - Treatment of 1-bromo-2-deuterio-2-phenylethane...Ch. 11.SE - Prob. 73APCh. 11.SE - Prob. 74APCh. 11.SE - In light of your answer to Problem 11-74, explain...Ch. 11.SE - Prob. 76APCh. 11.SE - Compound X is optically inactive and has the...Ch. 11.SE - When a primary alcohol is treated with...Ch. 11.SE - Prob. 79APCh. 11.SE - Amines are converted into alkenes by a two-step...Ch. 11.SE - The antipsychotic drug flupentixol is prepared by...
Knowledge Booster
Similar questions
- A student proposes the following reaction mechanism for the reaction in Model 6. Which step inthis mechanism is least favorable? Explain your reasoning.arrow_forwardShow which position of the 6 aromatic carbon is (are) the most electron rich by drawing detailed resonance delocalization with curved arrows. Conclude whether the molecule is ortho/para or meta director Predict the product of the following reaction and draw a detailed mechanism. Draw curved arrow to show the mechanism. Show two resonance structures for the intermediate. OCH 3 HNO3 H₂SO4arrow_forwardThe transformation below takes place by two distinct reactions. Intermediate A is formed in the first reaction and then this goes on to the product in the second reaction. Provide a complete curved-arrow mechanism for all steps of both reactions.arrow_forward
- Provide a detailed mechanism for each of the following reactions. a) b) c) سفی O=0 + NH,CH3 H₂O + CH3CH₂OH OH RiOH + ОН SOCI₂ H3O+ R CI + O=C H NHCH3 + H₂O SO₂ + HCIarrow_forwardUse the drop-down features to describe the mechanism of the given reaction. OH two Your answer is partially correct. one The first step of the mechanism is V curved arrow(s). The second step of the mechanism is curved arrow(s). The third step of the mechanism is protonation three M curved arrow(s). Hint H3O* Save for Later HO nucleophilic attack + ring opening ✔ deprotonation . This step requires . This step requires . This step requires Attempts: 1 of 2 used Submit Answer Fill in the Quest Multiste Ques Fill in t Ques Fill in Que Multi Que Multi Que Mult Qu Mul Sele Muarrow_forwardConsider the following reaction being performed with a low concentration. Think about what type of substitution mechanism will be favored, SN2 or SN1, and what product will result.arrow_forward
- Propose a mechanism for the following reactions. Use an appropriate organic chemistry software (e.g. chemsketch, chemdraw, etc.) to draw the structures and mechanism. Explain it.arrow_forwardThe following SN2 reaction gave J as a major product. Determine the structure of J. Explain your answer.arrow_forwardThe nucleophilic aromatic substitution reaction shown in the box proceeds via a benzyne intermediate and results in two organic products. What are structures of these products? CH3 NaNH2 ? CI liquid NH3 A) CH3 CH3 B) CH3 CH2NH2 CI -NH2 -NH2 NH2 CH3 CH3 D) CH3 CH3 -NH2 + -NH2 + -NH2 NH2 NH2 A В O Carrow_forward
- Provide major product and mechanism for the following reaction *most simplearrow_forwardThese two compounds shown go through SN1 substitution at a speed similar to some tertiary halides, even though the following compounds represent primary and secondary halides. Explain the enhanced reactivity via resonance structures and pushing arrows.arrow_forward3. SN2 reactions proceed with inversion of configuration, and SN1 reactions with loss of stereochemistry. However, the substitution reaction shown below proceeds with apparent retention of configuration. Provide a complete arrow pushing mechanism to plausibly explain this result. Br OH NaOH H₂O OH боло NaⒸarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning