
Interpretation:
The faster reactant is to be identified from given pairs of reactants with given reagent.
Concept introduction:
SN2 reaction:
Tosylated compound is reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
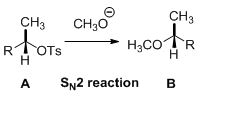
Given information:
a.
The given compound is shown below,
The SN2 displacement by I- on CH3Cl and CH3OTos
b.
The given compound is shown below,
The SN2 displacement by CH3COO- on bromoethane or on Bromo cyclohexane.
c.
The given compound is shown below,
The SN2 displacement on 2-bromopropane by CH3CH2O- or by CN-
d.
The given compound is shown below,
The SN2 displacement by CH ≡ C- on bromomethane in benzene or in acetonitrile
Trending nowThis is a popular solution!

Chapter 11 Solutions
Organic Chemistry
- 7В. (2 Draw the structure of the product, substrate or condition in the following reactions (should clearly indicate the stereochemistry). (а) OH (b) OsO vaHSO3 (c) 1) (sia)2 BH 2) NaOH/H,O2 H20 (d) HBr (2 equiv) Br, Br Hint: This is the only product formed (e) Br Br2 H20 OH (f)arrow_forwardNaOCH3 (S)-1-chloro-1,2-diphenylethane مين من cis-1,2-diphenylethene trans-1,2-diphenylethene + NaCl + HOCH 3 (a) Disregarding stereochemistry for the moment, provide a curved arrow mechanism for the transformation of the starting alkyl halide to one of the products.arrow_forward(i) OEt OEt 1) NaOEt 2) H3O* workup W 1) NaOEt 2) Br V Compound W can be synthesised in an excellent yield via an intramolecular Claisen reaction of 1,6-diester V. Draw the structure of product W and provide detailed reactions mechanisms to account for its formation. (ii) Draw the product X which would form upon the alkylation reaction of W with the reagents shown.arrow_forward
- 11. Plan syntheses of the following compounds. You may use the given starting material and any compound containing five or fewer carbons. (a) MeO MeO CN TOXXON anything with five or fewer carbons. CN (b) (c)arrow_forwardQ12. (1-bromoethyl)benzene 1 udergoes an elimination following an E1 mechanism. Fill in the following synthetic scheme by drawing the structure of intermediate 2 and product 3. The chemical formula of product 3 is provided as guidance. CH; RDS Br C3Hg E1 2 3arrow_forwardYour task is to convert 2-bromobutane to 1-butene in highest yield. Which reagents would you use? O KOH/H2O O KOH/CH,OH O CH3CH2ONA/CH3CH2OH O CH3ONA/CH3OH O (CH3)3COK/(CH3)3COHarrow_forward
- Q4. Discuss the reagents, conditions, and mechanism of the following reactions. a) Kolbe Reaction b) Williamson synthesis c) Reimer Tiemann reactionarrow_forwardProvide the structure(s) of the expected major organic product of the reaction shown. 1) Disiamylborane 2) H₂O₂, NaOH OI O II ||| O IV OV CH3CH₂C(CH3)2C=CH OH OH xx xo IVarrow_forwardOrder the compounds in each set according to their sn2-displacement reactivity:1-bromobutane; 1-bromo-2,2-dimethylpropane; 1-bromo-2-methylbutane; 1-bromo-3-methylbutane.arrow_forward
- What effect would you expect the following changes to have on the rate of the Sy2 reaction of 1-iodo-2-methylbutane with cyanide ion? (a) The CN- concentration is halved, and the l-iodo-2-methylbutane con- centration is doubled. (b) Both the CN- and the 1-iodo-2-methylbutane concentrations are tripled.arrow_forwardWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observed.(c) Use a Newman projection of the transition state to predict the major product of elimination of (2S,3R)-2-bromo-3-phenylbutanearrow_forwardWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observedarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





