
Interpretation:
The major
Concept introduction:
Elimination reaction:
An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
E1 elimination:
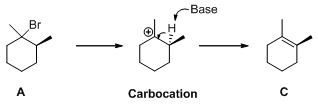
Saytzeff's Rule:
It is elimination reaction in which the formation of olefin on most hindered or highly substituted position.
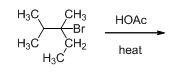
Given information:
The given compound is shown below,
Trending nowThis is a popular solution!

Chapter 11 Solutions
Organic Chemistry
- 3H. Put it all together. Predict the E1 products of the following reaction. Label alkenes as E/Z. 4. Predict the E1 products of the following reaction. Label akenes as E/Z.arrow_forward1 Predict the major alkene product of the following E1 reaction: H3C CH3 HOAC CH3CHCBr Неat CH2CH3arrow_forward7B What is the product of the following cleavage reaction and what is its stereochemistry? Me H Br H NaOEtarrow_forward
- Consider the following carbocation: Which of the following carbocations would result from its rearrangement? le lo A ΟΑ OB Save for Later > m B C Darrow_forward5. The E1 and E2 elimination reactions of this substrate lead to two different alkene isomer products (E or Z). Propose a mechanism for the E1 and E2 reactions and predict which isomer is the major product of each. Explain why each mechanism yields a different product. H3C, H H Br H₂O E1 VS. NaOH E2 E CH3 or Z CH3arrow_forwardTreatment of propadiene (an allene) with hydrogen bromide produces 2-bromopropene as the major product. This suggests that the more stable carbocation intermediate is produced by the addition of a proton to Br HBr. H2C=C=CH, H3C CH2 a terminal carbon rather than to the central carbon. Propadiene 2-Bromopropene (a) Draw both carbocation intermediates that can be produced by the addition of a proton to the allene. (b) Explain the relative stabilities of those intermediates. Hint: Draw the orbital picture of the intermediates and consider whether the CH, groups in propadiene are in the same plane.arrow_forward
- The following alkene is treated with one equivalent of NBS in CH2Cl2 in the presence of light to give bromination products. Please draw the structural formula for each product formed. Please it’s my last chance so it has to be right. Thank you!arrow_forwardChoose the diene that would produce the most stable carbocation intermediate upon treatment with HBr. A A В C D D E E A D B.arrow_forwardPropose a plausible mechanism for the following transformation: HO [H3O+] EtOH 19.55 The first three steps of the mechanism involve the formation of The first step is The second step is The third step is eTextbook and Media Save for Later Attemarrow_forward
- Predict the products of the following reaction, including stereochemistry. 1) OsO4 2) NaHSO, Write the mechanism and predict the products of the following reaction, including stereochemistry. Br2, H,Oarrow_forwardWittig Reaction - Examples Draw the mechanism and predict the product and its stereochemistry: Eto Br PPh3 nBuLi H Me 5 Mearrow_forwardE3C1-23s Complete the following reaction with all possible products, and determine which one is the major e 88 ОН K₂CO3arrow_forward
